Predict the Txy behavior for a mixture of ethanol (1) + 1-butanol (2) at 101.33 kPa using
Question:
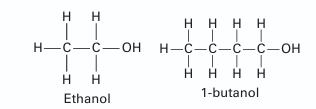
Predict the Txy behavior for a mixture of ethanol (1) + 1-butanol (2) at 101.33 kPa using the Peng Robinson equation of state Compare the pre dictions to the experimental data given in Table P12-21. Is the Peng-Robinson equation of state a reasonable model for this system at this state? Please explain.
![TABLE P12-21 Vapor-liquid equilibrium of ethanol (1) + 1-butanol (2) at 101.33 kPa. T[K] 390.75 388.15 385.65](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1698/4/0/0/477653b88dd58eb11698400472319.jpg)
Transcribed Image Text:
Η H II H=C=C-OH || Η Η Ethanol. Η Η Η Η H=C=C=C=C-OH ΙΙΙΙ Η Η Η Η 1-butanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
62+ Reviews
349+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Predict the Txy behavior for a mixture of n-hexane (1) + p-xylene (2) at 101.33 kPa using the Peng Robinson equation of state. Compare the predictions to the experimental data, as given in Table...
-
Predict the Txy behavior for a mixture of acetone (1) + 1-hexene (2) at 101.33 kPa using the Peng-Robinson equation of state. Compare the predictions to the experimental data given in Table P12-22....
-
Predict the Pxy behavior for a mixture of diethyl ether (1) + methanol (2) at 303.15 K using the Peng Robinson equation of state. Compare the predictions to the experimental data given in Table...
-
According to the 1980 revisions to the Copyright Act of 1976,_____. a. the owner of a copyrighted program is allowed to make multiplecopies or adaptations for use on other computers b. copyright...
-
Compare and contrast the role of transfer pricing in national versus international operations.
-
What business and technical skills are critical for todays business intelligence/analytics and information management professionals? As part of InformationWeeks 2013 U. S. IT Salary Survey, business...
-
19.11A Fallon Frodsham Limited manufactures and installs small prefabricated building structures that are sold to people who want to establish a home office using part of their gardens. Each...
-
Hirdt Co. uses the percentage-of-receivables basis to record bad debt expense and concludes that 2% of accounts receivable will become uncollectible. Accounts receivable are $400,000 at the end of...
-
It is the employee's responsibility to complete a new TD 1 form when ( 2 Points ) * a ) they now have an eligible dependent Apps b ) they no longer have tuition expenses c ) they no longer have an...
-
You work in a developing nation for a large chemical company. Your division works on refrigerants and foam-blowing agents. You have need to correlate a set of data for the trifluoromethane (1) +...
-
Predict the Pxy behavior for a mixture of 1,3-butadiene (1) + n-hexane (2) at 413.15 Kusing the Peng-Robinson equation of state. Compare the predictions to the experimental data given in Table...
-
Nathan Matthews conducted a test of controls where the tolerable deviation rate was set at 6 percent and the expected population deviation rate was 3 percent. Using a sample size of 150, Matthews...
-
Sparta Fashions owns four clothing stores, where it sells a wide range of women's fashions, from casual attire to formal wear. In addition, it rents formal wear and gowns for special occasions. At...
-
Problem 2-26 (Static) Complete the balance sheet using cash flow data LO 2-2, 2-3, 2-5, 2-6 Following is a partially completed balance sheet for Epsico Incorporated at December 31, 2022, together...
-
Consider the following potential events that might have occurred to Global Conglomerate on December30, 2018. For eachone, indicate which line items inGlobal's balance sheet would be affected and by...
-
An epidemiologist plans to conduct a survey to estimate the percentage of women who give birth. How many women must be surveyed in order to be 95% confident that the estimated percentage is in error...
-
Jamonit Ltd is a non-group employer which paid wages of $136,000 in the Northern Territory during March 2021. The company does not pay wages in any other state. Calculate the payroll tax payable in...
-
A steel beam is placed vertically in the basement of a building to keep the floor above from sagging. The load on the beam is 5.8 104 N, the length of the beam is 2.5 m, and the cross-sectional area...
-
What kind of financial pressures can an LBO cause?
-
A certain company makes televisions, stereo units, and speakers. Its parts inventory includes chassis, picture tubes, speaker cones, power supplies, and electronics. The inventory, required...
-
Plot the function y = 10(1 = e -x/4 ) over the interval 0 x x max , using a while loop to determine the value of x max such that y(x max ) = 9.8. Properly label the plot. The variable y represents...
-
Use a while loop to determine how many terms in the series 2 k , k = 1, 2, 3, . . . , are required for the sum of the terms to exceed 2000. What is the sum for this number of terms?
-
Some recent financial statements for Smolira Golf Corporation follow. 2020 2021 Assets Current assets Cash Accounts receivable Inventory SMOLIRA GOLF CORPORATION 2020 and 2021 Balance Sheets 2020...
-
Sunrise Coffee Shop, in an effort to streamline its accounting system, has decided to utilize a cash receipts journal. Record the following transactions for the first two weeks in March, total the...
-
You and your spouse wish to give each of your three grandchildren a yearly tax-free gift from your estate. If you give each of your three grandchildren the maximum amount allowed by law without...

Study smarter with the SolutionInn App


