We know that many binary mixtures contain an azeotrope, which means that the liquid and vapor phases
Question:
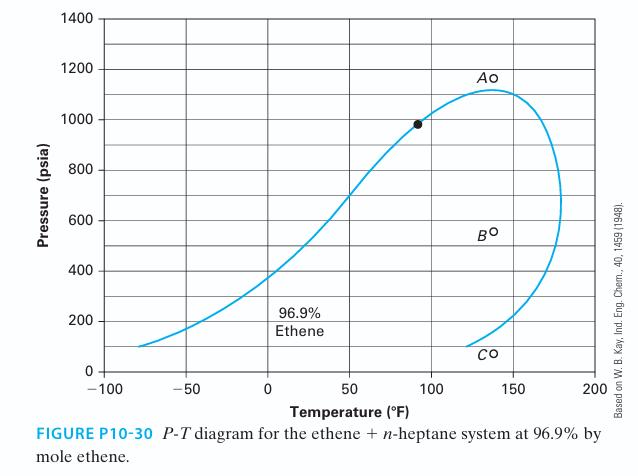
We know that many binary mixtures contain an azeotrope, which means that the liquid and vapor phases for the mixture have the same composition. Can a system have two azeotropes for a given pressure or temperature? If it exists, find a system and plot the data.
Transcribed Image Text:
Pressure (psia) 1400 1200 1000 800 600 400 200 0+ -100 -50 0 96.9% Ethene 100 Ao BO 50 Temperature (°F) FIGURE P10-30 P-T diagram for the ethene + n-heptane system at 96.9% by mole ethene. со 150 200 Based on W. B. Kay, Ind. Eng. Chem., 40, 1459 (1948).
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
The overall goal of this problem is to compute the PV and PT equilibrium diagramsfor a single component fluid described by the van derWaals equation of state. Let us recall the key things we need to...
-
Vapor?liquid equilibrium data for mixtures of acetone (A) and ethanol at 1 atm are given in the following table: (a) Use the given data to construct a Txy diagram for this system. (b) A thermocouple...
-
A binary mixture of mole fraction zj is flashed (o conditions T and P, Fur one of the following determine: the equilibrium mole fractions x1 and y1 of the liquid and vapor phases formed, the molar...
-
Q8 Question: 9 A small particle of mass m moving inside a heavy, hollow and straight tube along the tube axis undergoes elastic collision at two ends. The tube has no friction and it is closed at one...
-
Jody Hunter and Jim Boling, two managers at Georgia- Pacific Corporation, a paper manufacturer, disagree about continuing the companys membership in Business for Affordable Medicine (BAM), a...
-
Suppose you intend to draw a sample of 100 residents from the large population of residents living in Blanchard County, Mississippi. You plan to compute the mean age (x) in the sample. The most...
-
What were you trying to achieve?
-
Reboot Inc. provides computer repair services for the community. Ashley DaCostas computer was not working, and she called Reboot for a home repair visit. The Reboot Inc. technician arrived at 2:00 p....
-
please asap thankyou Martel Corporation is a manufacturing business that produces corporate merchandise. For production of one their main product that is stainless steel tumbler, they utilise a...
-
Figure P10-30 is a P-T diagram for the ethene + n-heptane system at 96.9% by mole ethene (Kay, 1948). Please answer the following questions based on this figure. Note that the in the figure...
-
A separation stream off the main reactor effluent contains almost exclusively ethyl benzene, benzene, and toluene at 1 bar and 100C. You determine that the stream flow rate is made up of 34 kg/s of...
-
On November 21, 2011, a fire at Hodge Company's warehouse caused severe damage to its entire inventory of Product Tex. Hodge estimates that all usable damaged goods can be sold for $12,000. The...
-
The domain of chemical reaction engineering consists of all chemical transformations (and that includes biological) of starting materials, derived from non-renewable and renewable resources, into a...
-
What is judgement sampling?Explain with a suitable example.
-
What is convenience sampling?Explain with a suitable example.
-
Customer Distribution Channels (all amounts in thousands of U.S. Dollars) Wholesale Customers Retail Customers Total Total N. America S. America Total Wholesale Wholesaler Wholesaler Retail Green...
-
MA Assignment 3 Motor Tyres manufactures one size of tyre in each of its production lines. The following information relates to one production line for the most recent period. The company uses the...
-
In an aviation test lab, pilots are subjected to vertical oscillations on a shaking rig to see how well they can recognize objects in times of severe airplane vibration. The frequency can be varied...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
The mechanical work W done in using a force F to push a block through a distance D is W ! FD. The following table gives data on the amount of force used to push a block through the given distance...
-
Plane A is heading southwest at 300 mi/hr, while plane B is heading west at 150 mi/hr. What are the velocity and the speed of plane A relative to plane B?
-
The following table shows the hourly wages, hours worked, and output (number of widgets produced) in one week for ve widget makers. Use MATLAB to answer these questions: a. How much did each worker...
-
Lina Center for Performing Arts is a not-for-profit organization. The center began operations on January 1, 2021 with $135,000 of temporarily restricted assets. The amounts are restricted for the...
-
Singer Company has a line of credit with United Bank. Singer can borrow up to $307,000 at any time over the course of the Year 1 calendar year. The following table shows the prime rate expressed as...
-
please need urgent help with these 2 journal entries On February 3 , a company provides services on account for $26,500, terms 2/10,n/30. On February 9 , the company recelves payment from the...

Study smarter with the SolutionInn App


