Repeat Prob. 1643 for liquid oxygen, which has a boiling temperature of - 183°C, a heat of
Question:
Repeat Prob. 16€“43
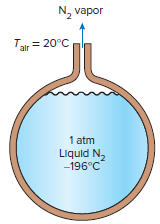
The boiling temperature of nitrogen at atmospheric pressure at sea level (1 atm) is -196°C. Therefore, nitrogen is commonly used in low-temperature scientific studies since the temperature of liquid nitrogen in a tank open to the atmosphere remains constant at -196°C until the liquid nitrogen in the tank is depleted. Any heat transfer to the tank results in the evaporation of some liquid nitrogen, which has a heat of vaporization of 198 kJ/kg and a density of 810 kg/m3 at 1 atm.
Consider a 4-m-diameter spherical tank initially filled with liquid nitrogen at 1 atm and -196°C. The tank is exposed to 20°C ambient air with a heat transfer coefficient of 25 W/m2·K. The temperature of the thin-shelled spherical tank is observed to be almost the same as the temperature of the nitrogen inside. Disregarding any radiation heat exchange, determine the rate of evaporation of the liquid nitrogen in the tank as a result of the heat transfer from the ambient air.
Step by Step Answer:

Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala





