At 298 K, f G[CO(g)] = -137.2 kJ mol -1 and K = 6.5 x 10
Question:
At 298 K, ΔfG°[CO(g)] = -137.2 kJ mol-1 and K = 6.5 x 1011 for the reaction CO(g) + Cl2(g) ⇌ COCl2(g). Use these data to determine ΔfG°[COCl2(g)], and compare your result with the value in Appendix D.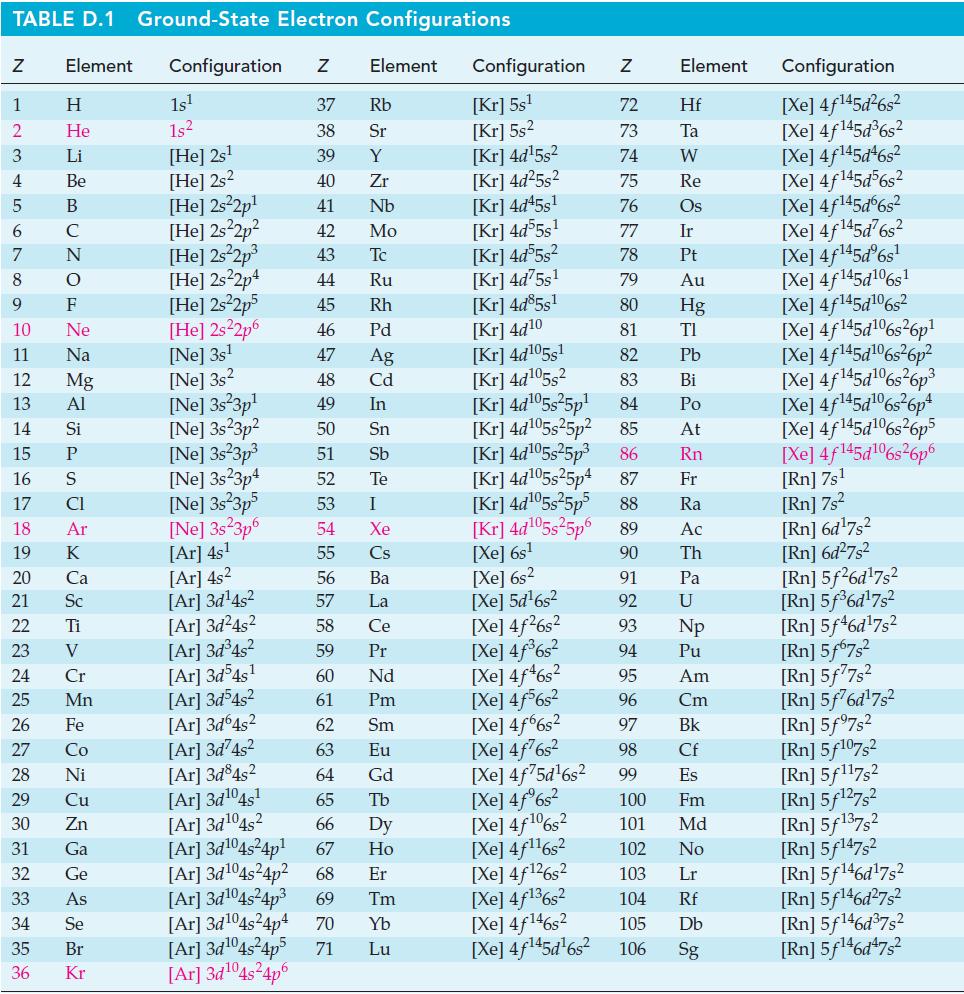
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZON SJE ZE> 0 ≤ 2 8 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 [Ar]3d²4s² 63 [Ar] 3d845² 64 [Ar]3d¹04s¹ 65 [Ar] 3d¹04s2 66 67 [Ar]3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Er [Ar] 3d¹04s²4p³ 69 Tm 70 Yb Lu [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² [Ar] 3d²4s² [Ar] 3d³4s² [Ar]3d54s¹ [Ar] 3d³4s² [Ar] 3d64s² Element [Ar] 3d¹04s²4p4 [Ar] 3d¹04s²4p5 71 [Ar] 3d¹04s²4p6 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine fG for the reaction COg Cl2g COCl2g using the given information you can use the relatio...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
In Chapters 2 (Section 2.8) and 3 (Section 3.7) we analyzed the risk and return of the Orange County portfolio, using the building blocks of zero coupon bonds and duration in this exercise, we...
-
A tabulation of more precise thermodynamic data than are presented in Appendix D lists the following values for H 2 O(l) and H 2 O(g) at 298.15 K, at a standard state pressure of 1 bar. (a) Use these...
-
The United Arab Emirates (UAE) is unique in that expatriates constitute more than 80 per cent of the population. As the country continues to grow and accept foreigners at an astounding pace, Emiratis...
-
Fuqua Companys sales budget projects unit sales of part 198Z of 10,000 units in January, 12,000 units in February, and 13,000 units in March. Each unit of part 198Z requires 4 pounds of materials,...
-
In Figure l 7.6b, suppose that for some point P on the screen r 1 = 5,002,248.5A and r 2 = 5,002,25 l.5A, where A is the wavelength of the light. The interference at point P is A. Constructive. B....
-
Consider a high school with which you are familiar. How can knowledge management at this high school benefit from information technologies? What kinds of technologies does it currently use, and how...
-
Marcelino Co.'s March 31 inventory of raw materials is $80AX/0. Raw materials purchases in April are $500,000, and factory payroll cost in April is $363,000. Overhead costs incurred in April are:...
-
Top New Features in Sure Cuts A lots Top New Features in Sure Cuts A Lot 5 - YouTube Ch 3: Homework QS 3-4 Concepts of adjusting entries LO P1, P2, P3, P4 9.09 During the year, a company recorded...
-
At 1000 K, an equilibrium mixture in the reaction CO 2 (g) + H 2 (g) CO(g) + H 2 O(g) contains 0.276 mol H 2 0.276 mol CO 2 , 0.224 mol CO, and 0.224 mol H 2 O. (a) What is K at 1000 K? (b)...
-
Why is r G such an important property of a chemical reaction, even though the reaction is generally carried out under nonstandard conditions?
-
The pole supports a traffic light of weight W. Using Cartesian vectors; determine the moment of the weight of the traffic light about the base of the pole at A. Given: W = 22 lb a = 12 ft θ =...
-
what ways do mindfulness-based stress reduction (MBSR) techniques contribute to reducing stress, and what are their limitations ?
-
How do search engines understand "user experience" to evaluate organic rankings?
-
How do you think a risk manager might work through a difference of opinion with a unit manager? Explain in details.
-
How do transnational organizations and agreements influence national sovereignty and political autonomy ?
-
How do individuals reconcile the tension between rational deliberation and emotional impulses when making consequential decisions amidst volatile environments, and to what extent does the phenomenon...
-
Find the input impedance of the circuit in Fig. 13.99 using the concept of reflected impedance. Obtain the input impedance by replacing the linear transformer by its T equivalent. j10 j40 25 8 2...
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
FIFO method (continuation of 17-30). Do Problem 17-30 using the FIFO method of process costing. Explain any difference between the cost per equivalent unit in the Assembly Department under the...
-
Transferred-in costs, weighted average method (related to 17-30 to 17-32). Larsen Company, as you know, is a manufacturer of car seats. Each car seat passes through the Assembly Department and...
-
Transferred-in costs, FIFO method (continuation of 17-33). Refer to the information in Problem 17-33. Suppose that Larsen Company uses the FIFO method instead of the weighted-average method in all of...
-
In which transaction cycle would information for retiring long-term debt be most likely to pass between internal and external accounting information systems. Select one: A. the financing cycle B. the...
-
What is the purpose of tests of controls? With reference to the three (3) main risk components of the Audit Risk Model, explain the circumstances where it is NOT appropriate for the Auditor to test...
-
50 If at the end of each month you save 300 for 40 years and earn 8 % annually, what is the Future Value of your savings

Study smarter with the SolutionInn App


