Describe a hybridization scheme for the central S atom in the molecule SF 4 that is consistent
Question:
Describe a hybridization scheme for the central S atom in the molecule SF4 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the S atom are involved in overlaps, and which are occupied by lone-pair electrons?
Table 10.1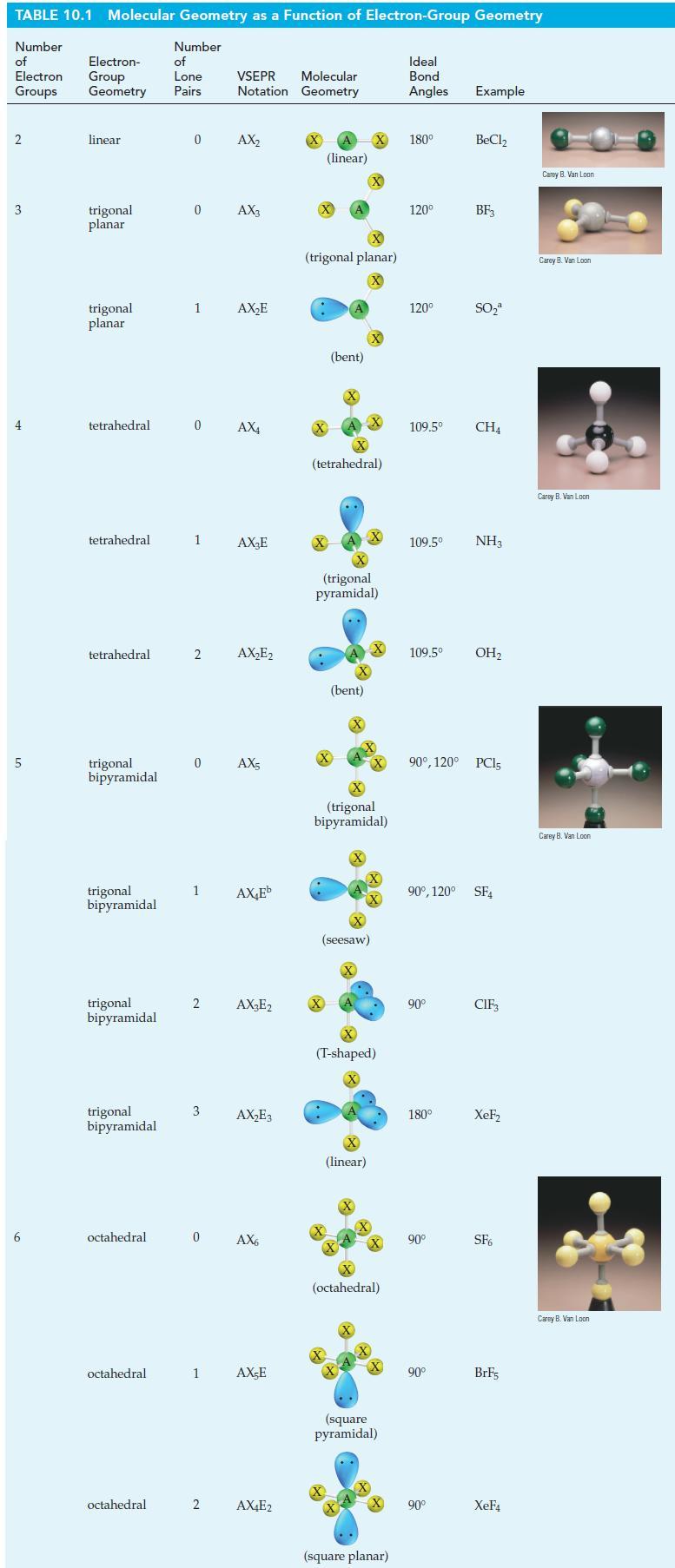
Transcribed Image Text:
TABLE 10.1 Molecular Geometry as a Function of Electron-Group Geometry Number of Electron Groups 2 3 4 5 6 Electron- Group Geometry linear trigonal planar trigonal planar tetrahedral tetrahedral tetrahedral trigonal bipyramidal trigonal bipyramidal trigonal bipyramidal trigonal bipyramidal octahedral octahedral octahedral Number of Lone Pairs 0 0 1 1 2 0 AX4 1 2 3 Molecular VSEPR Notation Geometry 0 AX₂ AX3 0 AX5 2 AX₂E AX₂E AX₂E₂ AX₂Eb AX₂E2 AX₂E3 AX6 1 AX-E AX4E2 (linear) X (trigonal planar) X X (bent) (tetrahedral) .. X (trigonal pyramidal) (bent) X X (seesaw) X X (trigonal bipyramidal) X (linear) (T-shaped) X X +4+ (octahedral) X X (square pyramidal) (square planar) Ideal Bond Angles Example 180⁰ 120⁰ 120⁰ 109.5⁰ 109.5⁰ 109.5⁰ NH3 90° 180° BeCl₂ 90° BF3 90⁰, 120° PC15 90° SO₂ 90⁰, 120° SF4 90° CH4 OH₂ CIF₁ XeF₂ SF6 BrF5 XeF4 Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Describe a hybridization scheme for the central Cl atom in the molecule ClF 3 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the Cl atom are involved in...
-
In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (a) What is the best choice of hybridization scheme for the atoms of ozone? (b) For one of the resonance...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Consider the nutrition problem in Example 1 of Section 3.3. Solve the problem by the simplex method, and then determine the optimal quantities of soybeans and rice in the diet, and the new cost, if...
-
Amos orally agrees to hire Elizabeth for an eight-month trial period. Elizabeth performs the job magnificently, and after several weeks Amos orally offers Elizabeth a six-month extension at a salary...
-
Why do you think business-to-consumer (B2C) transactions are less likely to develop on the internet, both domestically and globally?
-
Auditors would argue that in many instances the exercise of hindsight is used to identify negligence in circumstances where the issue was in fact one of judgement. Discuss with particular reference...
-
Which organizational form would be best for the following corporate strategies? a. Developing, manufacturing, and marketing many diverse but interrelated technological products and materials b....
-
Which steps used to complete a work sheet are in the correct order? O enter the adjusted balances, calculate profit or loss, enter the adjustments O enter the adjustments, enter the adjusted...
-
1. Suppose you wanted to predict Winnings ($) using only the number of poles won (Poles), the number of wins (Wins), the number of top five finishes (Top 5), or the number of top ten finishes (Top...
-
(A) Melamine is a carbonhydrogennitrogen compound used in the manufacture of adhesives, protective coatings, and textile finishing (such as in wrinkle-free, wash-and-wear fabrics). Its mass percent...
-
Propose a plausible Lewis structure, geometric structure, and hybridization scheme for the ONF molecule.
-
Compute the first partial derivatives of the following functions. g(x, y, z) = 4x - 2y 2z - 6x - 3z
-
Newfoundland Hapset will be remitted to _____
-
The Z Energy Corp. has a new investment opportunity that generates cash flows of $6 million per year (in expectation) forever. The managers of Z Energy are not sure what the required rate of return...
-
Define an interface TwoStrings Oper declaring a function apply which takes two strings and returns a string. Then, define four classes implementing this interface, where the operation on strings...
-
Noeleen AutoMall, Ltd. recently completed an initial public offering(IPO) for$23,000,000 by listing its common shares on the New York Stock Exchange. Prior to itsIPO, Noeleen was a privately held...
-
Process Costing- increased units, FIFO method Answer in good form 25 26 Illustrative Problem-Cost of Production Report using Treatment by Neglect Dept 1-100% of materials are added at the beginning....
-
Write structures for each of the following isomers and arrange them in order of decreasing boiling point: 3-methoxy-1-propanol, 1,2-dimethoxyethane, and 1,4-butanediol.
-
For a Poisson process of rate , the Bernoulli arrival approximation assumes that in any very small interval of length , there is either 0 arrivals with probability 1- or 1 arrival with probability ....
-
Credit Instruments Describe each of the following: a. Sight draft b. Time draft. c. Bankers acceptance. d. Promissory note. e. Trade acceptance.
-
Trade Credit Forms in what form is trade credit most commonly offered? What is the credit instrument in this case?
-
Receivables Costs what costs are associated with carrying receivables? What costs are associated with not granting credit? What do we call the sum of the costs for different levels of receivables?
-
Calculate the present value of cash flows, 1500 in the years 1, 2, 3, and 4, then grows at 2% every year, using 10% discount rate. Round and write up to two decimals (e.g., 100.00). No characters...
-
What are the two formats that companies can use to present their Statement of Comprehensive Income?
-
Problem 15-19 (Algo) Initial direct costs; sales-type lease (LO15-2, 15-7) Bidwell Leasing purchased a single-engine plane for its fair value of $834,564 and leased it to Red Baron Flying Club on...

Study smarter with the SolutionInn App


