Estimate the normal boiling point of bromine, Br 2 in the following way: Determine vap H
Question:
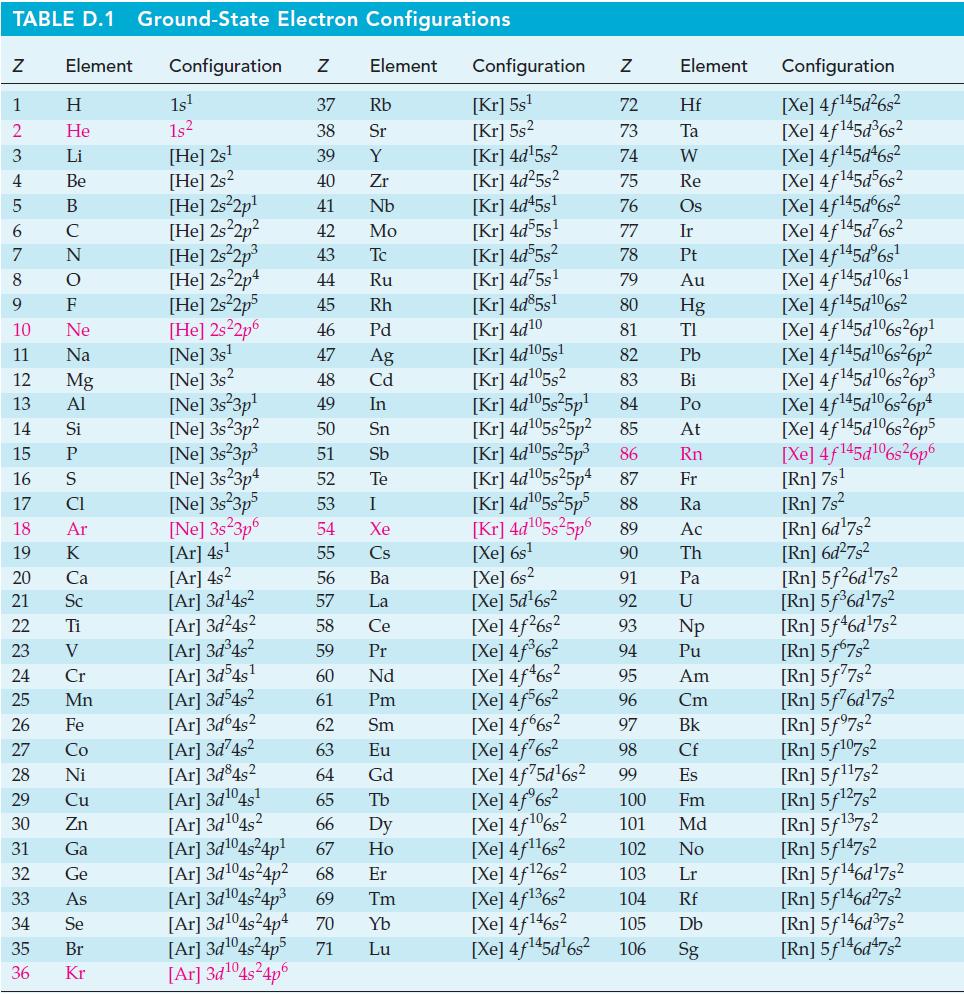
Estimate the normal boiling point of bromine, Br2 in the following way: Determine ΔvapH° for Br2 from data in Appendix D. Assume that ΔvapH° remains constant and that Trouton’s rule is obeyed.
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 6 7 8 9 5 B 10 11 12 13 14 15 16 17 18 19 20 25 27 29 HIG&LUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 2 2 5 2 30 Η 31 He 32 Li 33 Be C F Ne Na Mg Al 21 Sc 22 Ti 23 V 24 Si P CI Ar 26 Fe K Ca 28 Ni Cr Mn Co Cu Zn Ga Ge As 34 Se 35 Br 36 Kr 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² Element 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb Te 52 53 54 55 56 57 58 59 60 61 62 63 64 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd [Ar] 3d²4s² [Ar] 3d³4s² [Ar] 3d54s¹ [Ar] 3d³4s² [Ar]3d64s² [Ar] 3d74s² [Ar] 3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 65 Tb 66 67 [Ar] 3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Dy Ho Er [Ar] 3d¹04s²4p³ 69 Tm [Ar] 3d¹04s²4p4 70 Yb [Ar]3d¹04s²4p5 71 Lu [Ar]3d¹04s²4p6 Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [kr] 4d¹05s²5p¹ [kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f145d6s2 [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
1 Determine vapH for Br2 from data in Appendix D From Appendix Dwe can find that the ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Trouton's rule states that for many liquids at their normal boiling points, the standard molar entropy of vaporization is about 88 J/mol-K. (a) Estimate the normal boiling point of bromine, Br2, by...
-
The normal boiling point of hexane is 69.0C. Estimate (a) Its enthalpy of vaporization and (b) Its vapour pressure at 25C and 60C.
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
Use Lagrange multipliers to find the maximum area of a rectangle inscribed in the ellipse (Figure 15): (-x,y) (x, y) 2 a2 b2 + J2 = 1 (x, y) (x, y) X
-
In what situation does a company place the greatest focus on its target cost? How is the target cost determined?
-
Steven had a taxable estate of $8,850,000 when he died in 2013. If he had no prior taxable gifts, what is his net estate tax liability?
-
Persistent Earnings Identify each of the following items as either (P) persistent, or (T) transitory. a. Sale of merchandise. b. Settlement of a lawsuit. c. Interest income. d. Payment to vendors. e....
-
As a financial analyst, you have just been handed the 2012 financial report of Firm A, a large, global pharmaceutical company. Firm A competes in both traditional pharmaceutical products and in...
-
On your own words , please! Describe and explain briefly the question below; Kindly give samples! 1. What is system safety? 2. What is the definition of a hazard?
-
Pentane is one of the most volatile of the hydrocarbons in gasoline. At 298.15 K, the following enthalpies of formation are given for pentane: f H[C 5 H 12 (l)] = -173.5 kJ mol -1 ; f H[C 5 H 12...
-
Calculate the entropy change, S, for the following processes. If necessary, look up required data in Appendix D. (a) The pressure of one mole of O 2 (g) is increased from P to 2P at 298 K. (b) The...
-
At constant temperature, the volume of a given mass of a gas is inversely proportional to the pressure. This is known as (a) Avogadro's law (b) Charles' law (c) Boyle's law (d) none of these.
-
3. What will happen to stock price if the number of years in the forecast period is changed, and you add 10 years
-
What are the implications of cultural competence and cultural intelligence for fostering inclusive work environments that celebrate differences and promote collaboration across diverse teams?
-
Define leverage and explain the meaning of procyclical leverage. Intuitively explain how investment banks can obtain this type of leverage.
-
What are the ethical implications of employing coercive tactics, such as ultimatums or sanctions, in conflict resolution efforts, and how can ethical dilemmas be navigated to uphold moral integrity ?
-
Service cost $207,000 Contribution to the plan 268,000 Prior service cost amortization 34,000 Actual and expected return on plan assets 127,000 Benefits paid 225,000 Plan assets at January 1, 2025...
-
Pizza sales and price, part 2. For the data in Exercise 31, the average Sales was 52,697 pounds (SD = 10,261 pounds), and the correlation between Price and Sales was = 0.547. If the Price in a...
-
3M Company reports the following financial statement amounts in its 10-K report: a. Compute the receivables, inventory, and PPE turnover ratios for both 2018 and 2017. (Receivables turnover and...
-
Harris Fabrics computes its predetermined overhead rate annually on the basis of direct labor hours. At the beginning of the year it estimated that its total manufacturing overhead would be S 134,000...
-
Lamed Corporation recorded the following transactions for the just completed month. a. $80,000 in raw materials were purchased on account. b. $71,000 in raw materials were requisitioned for use in...
-
Luthan Company uses a predetermined overhead rate of $23.40 per direct labor-hour. This predetermined rate was based on 11,000 estimated direct labor-hours and $257,400 of estimated total...
-
Flag question: Question 16 Two years ago Calima Financial entered a fixed for floating rate swap to receive fixed payments and pay floating rate payments. In this swap, payments are made every...
-
cEthel is interested in two methods of note-taking strategies and the effect of these strategies on the overall GPAs of college freshmen. She believes that men would benefit most from Method 1, while...
-
1. A project requires an initial cash outlay of $90,000 and is expected to generate $35 000 at the end of year 1, $43 000 at the end of year 2 and $40 000 at the end of year 3, at which time the...

Study smarter with the SolutionInn App


