In the drawing it shown that, 1.00 g H 2 (g) at 300 K is maintained at
Question:
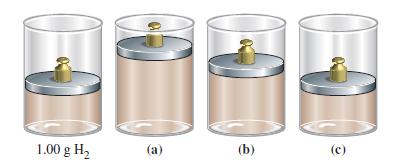
In the drawing it shown that, 1.00 g H2(g) at 300 K is maintained at 1 atm pressure in a cylinder closed off by a freely moving piston. Which sketch, (a), (b), or (c), best represents the mixture obtained when 0.50 g H2(g) is added and the temperature is reduced to 275 K? Explain your answer.
Transcribed Image Text:
1.00 g H₂ (a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The correct answer is b When 050 g H2g is added to the cylinder the total ...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Can you think of a soft drink besides Pepsi that has the potential to outsell Coca Cola Explain your answer How can Pepsi compete with Coke Is there a market niche the company has not targeted that...
-
In the drawing below, 1.00 g H 2 (g) is maintained at 1 atm pressure in a cylinder closed off by a freely moving piston. Which sketch, (a), (b), or (c), best represents the mixture obtained when 1.00...
-
A gas mixture consists of 320 mg of methane, 175 mg of argon, and 225 mg of neon. The partial pressure of neon at 300 K is 8.87 kPa. Calculate (a) The volume and (b) The total pressure of the mixture.
-
Use the percentages in Figure 7.3 and the values in Figure 7.4 to answer the following questions: What percentage of SAT takers score between 500 and 600? Figure 7.3 Normal Curve Percentages, to Two...
-
Three students share a house. Having better things to do than clean house, they hire someone to come in and clean once each week. How should they share the costs of the housekeeper? One simple...
-
In a survey conducted by CareerBuilder.com, employers were asked if they had ever sent an employee home because they were dressed inappropriately (June 17, 2008, www.careerbuilder.com). A total of...
-
The following information is taken from the annual report of The Coca-Cola Company: Using this information, calculate the accounts payable turnover ratio and the days payable period for 2015 and...
-
1. If the variable cost per unit goes up, Contribution margin Break-even point a. Increases increases. b. Increases decreases. c. Decreases decreases. d. Decreases increases. e. Decreases remains...
-
https://www.cbc.ca/news/canada/north/parks-canada-sexual-harassment-1.6238820 KINDLY ASSIST WITH A DETAILED SUMMARY OF THE CASE IN THE LINK ABOVE
-
A 2.35 L container of H 2 (g) at 762 mmHg and 24 C is connected to a 3.17 L container of He(g) at 728 mmHg and 24 C. After mixing, what is the total gas pressure, in millimeters of mercury, with the...
-
A gas cylinder of 53.7 L volume contains N 2 (g) at a pressure of 28.2 atm and 26 C. How many grams of Ne(g) must we add to this same cylinder to raise the total pressure to 75.0 atm?
-
Mr. and Mrs. Gamble paid $53,000 for a corporate bond with a $50,000 stated redemption value. They paid the $3,000 premium because the bonds annual interest rate is higher than the market interest...
-
What is the problems with selecting a jury pool that reflects our racially diverse populations? What are two ways to improve jury selection to ensure that a criminal defendant has a jury that can be...
-
What is the difference between evaluative and facilitative mediation? What approach would you take if were a mediator? Which approach would you prefer a mediator take if you were an advocate...
-
Tonic Inc. is a manufacturing company. Last year, the company generated $75 million in EBIT. The company also had $50 million in capital expenditures and reported $20 million in depreciation and...
-
A scale reads 264 N when a piece of iron is hanging from it. What does it read (in N) when it is lowered so that the iron is submerged in water? Please give your answer in N 2. A boat (with a flat...
-
A block of mass 3kg is given an initial velocity c= 5.0 m/s up a frictionless incline of angle 24 o . How far up along the incline does the block slide before coming to a momentary rest? State your...
-
What is the underlying philosophy of the Sherman Act with respect to the role of competition versus monopoly in promoting public welfare? Discuss.
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
Archer Inc. issued $4,000,000 par value, 7% convertible bonds at 99 for cash. If the bonds had not included the conversion feature, they would have sold for 95. Prepare the journal entry to record...
-
Petrenko Corporation has outstanding 2,000 $1,000 bonds, each convertible into 50 shares of $10 par value common stock. The bonds are converted on December 31, 2010, when the unamortized discount is...
-
Pechstein Corporation issued 2,000 shares of $10 par value common stock upon conversion of 1,000 shares of $50 par value preferred stock. The preferred stock was originally issued at $60 per share....
-
You are considering a stock investment in one of two firms (LotsofDebt, Incorporated and LotsofEquity, Incorporated), both of which operate in the same industry. LotsofDebt, Incorporated finances its...
-
Blooper Industries must replace its magnoosium purification system. Quick & Dirty Systems sells a relatively cheap purification system for $16 million. The system will last 4 years. Do-It-Right sells...
-
3 2 points Problem 7-1 (LO7.1) Based on the following data, calculate the items requested: Rental Costs Annual rent Buying Costs eBook Hint Print References $ 8,080 Annual mortgage payments Insurance...

Study smarter with the SolutionInn App


