Write K sp expressions for the following equilibria. For example, for the reaction AgCl(s) Ag +
Question:
Write Ksp expressions for the following equilibria. For example, for the reaction AgCl(s) ⇌ Ag+(aq) + Cl-(aq), Ksp = [Ag+][Cl-].
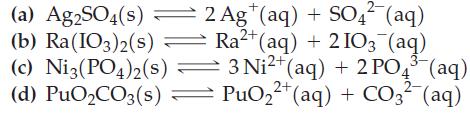
Transcribed Image Text:
(a) Ag2SO4(s) (b) Ra(103)2(s) 2 Ag+ (aq) + SO4² (aq) Ra²+ (aq) + 2103 (aq) (c) Ni3(PO4)2(s) — 3 Ni²+ (aq) + 2 PO4³(aq) 2+ 2- (d) PuO₂CO3(s) = PuO₂²+ (aq) + CO3²- (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The solubility product constant Ks...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) K sp for AgCl = 1.8 x 10 -10 . What would be the measured E cell for the voltaic cell in Example 19-11 if the contents of the anode half-cell were saturated AgCl(aq) and AgCl(s)? Example 19-11...
-
Write regular expressions for the following languages 1. Language of all words that start with x or y and ends with x or z. 2. Language of all words that do not have x or y at the third position. 3....
-
Write solubility product expressions for the following compounds. a. Mg(OH)2 b. SrCO3 c. Ca3(AsO4)2 d. Fe(OH)3
-
Selected accounts of Holly Company are shown as follows. Instructions After analyzing the accounts, journalize (a) The July transactions (b) The adjusting entries that were made on July 31. Supplies...
-
The following financial statements are for Troi Company. Consider the following additional information: (a) All accounts payable relate to inventory purchases. (b) Property, plant, and equipment sold...
-
Use a MATLAB built-in function to numerically solve: dy/dx = x2 + x3ey/4 for 1 < x < 5 with y(1) = 1
-
What are the motivational opportunities of alternative work schedules?
-
1. Is such a loosely organized firm likely to be as effective as a firm that defines jobs more precisely and monitors performance more closely? What are the advantages and the limitations of the...
-
Gmail D2L Brightspace TRIGONOMETRIC FUNCTIONS Coterminal angles Answer the following. (a) Find an angle between 0 (b) Find an angle between 0
-
Write solubility equilibrium equations that are described by the following K sp expressions. For example K sp = [Ag + ][Cl] represents AgCl(s) Ag + (aq) + Cl(aq). 3+ (a) Ksp = [Fe+][OH-] (b) Ksp =...
-
From the observations listed, estimate the value of E for the half-cell reaction M 2+ (aq) + 2 e M(s). (a) The metal M reacts with HNO 3 (aq), but not with HCl(aq); M displaces Ag + (aq), but not Cu...
-
Assume that in preparing the cost of goods manufactured schedule, several errors were made. Raw materials purchases, sales revenue and factory depreciation should have been $106,400, $584,000 and...
-
Physical Units Beginning WIP 191 New units started 463 Ending WIP 56 *Beginning WIP was 100% complete for direct materials, and 40% complete for conversion costs **Ending WIP was 100% complete for...
-
Physical Units Work in Process, Jan 1* Started during the year 40 Direct Materials Conversion Costs $6,000 $700 43 Work in Process, Dec 31** 26 Total cost added in the year $16,000 $6,400 Beginning...
-
The purpose of this problem is to determine the performance of residential vapor compression heat pump operating in the heating mode using refrigerant R134a. The heat pump system has a fixed speed...
-
Research scholarships that fit your personal profile and estimate your potential financial aid. Then you will analyze the results of your research and explain your plan for achieving all the aid you...
-
Part A 1. Tear part of a piece of paper into small bits of various sizes. Take a plastic pen and bring it close to the bits of paper. Can you lift the bits of paper by touching them with the pen? 2....
-
A certain model of remote-control Stanley garage door opener has nine binary (off/on) switches. The homeowner can set any code sequence. (a) How many separate codes can be programmed? (b) If you try...
-
A sprinkler head malfunctions at midfield in an NFL football field. The puddle of water forms a circular pattern around the sprinkler head with a radius in yards that grows as a function of time, in...
-
Dilts Manufacturing Inc. had sales of $1,800,000 for the first quarter of 2010. In making the sales, the company incurred the following costs and expenses. Prepare a CVP income statement for the...
-
Gore Companys fixed overhead costs are $3 per unit, and its variable overhead costs are $8 per unit. In the first month of operations, 50,000 units are produced, and 47,000 units are sold. Write a...
-
Montana Company reports the following total costs at two levels of production. Classify each cost as variable, fixed, or mixed. 5,000 Units 10,000 Units Indirect labor Property taxes Direct labor...
-
ces Prepare a horizontal analysis of both the balance sheet and income statement. Complete this question by entering your answers in the tabs below. Analysis Bal Analysis Inc Sheet Stmt Prepare a...
-
XYZ Corporation presents in its accounting books the following items related to its income: Sale income $2,000,000 Cost of Goods Sold (1,300,000) Savings account interest income 1,000 New York City...
-
Crane Company Division B recorded sales of $71,700, variable cost of goods sold of $49,600, variable selling expenses of $19,300, and fixed costs of $12,400, creating a loss from operations of...

Study smarter with the SolutionInn App


