In the fabrication of semiconductors, one step is the formation of additional gallium arsenide (GaAs) on the
Question:
In the fabrication of semiconductors, one step is the formation of additional gallium arsenide (GaAs) on the surface of a GaAs wafer. To accomplish this, gaseous tri-methyl gallium (TMG) and arsine flow in a carrier gas (usually hydrogen) over the solid and react on the solid surface at a temperature of 900K to produce the solid GaAs and gaseous methane. The unreacted TMG and arsine and the newly produced methane flow out in the outlet gas.
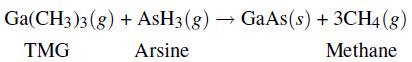
This process can be treated as a steady-state process wherein the deposition of the solid is treated as an outlet stream of pure solid GaAs. For such a process, the following are given:
 where the quantity in the solid “out” indicates that we wish to deposit 7 mg/min of GaAs. Also, we have assumed that the flow rate of the gas stream doesn’t change significantly.
where the quantity in the solid “out” indicates that we wish to deposit 7 mg/min of GaAs. Also, we have assumed that the flow rate of the gas stream doesn’t change significantly.
What is the concentration of methane in the outlet gas?
Step by Step Answer:

Introduction To Chemical Engineering Tools For Today And Tomorrow
ISBN: 9780470885727
5th Edition
Authors: Kenneth A. Solen, John N. Harb





