Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.80, initially at 80C and 100 kPa. Describe
Question:
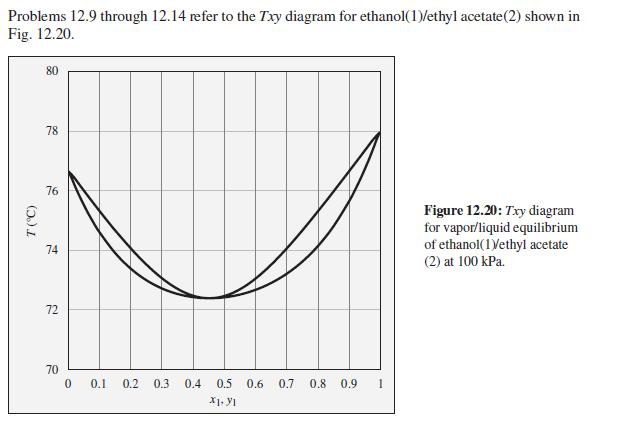
Consider an ethanol(1)/ethyl acetate(2) mixture with x1 = 0.80, initially at 80°C and 100 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to 70°C.
Transcribed Image Text:
Problems 12.9 through 12.14 refer to the Txy diagram for ethanol(1)/ethyl acetate(2) shown in Fig. 12.20. 80 78 76 Figure 12.20: Txy diagram for vapor/liquid equilibrium of ethanol(1Vethyl acetate 74 (2) at 100 kPa. 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1 X1. YI (5.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
A temperaturecomposition phase diagram for the given mixture is required to determine the evolution of phases and phase compositions as the temperatur...View the full answer

Answered By

Gitanjali Singh
I'm a detail-oriented Economists and Accountant with three year effectively maintaining accurate accounting information for large and small-scale financial organizations. History working as part of financial team to manage diverse financial functions, tax management and reporting.
In my experience, tutoring tends to be more goal oriented and standardized where I'm leading lessons and reviewing materials with a focus on helping the student to pass the test. Hence I love tutoring and I get a sense of achievement from seeing my students grow and develop as individuals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
Jake's office building is destroyed by a fire on September 15, 2022. Jake receives insurance proceeds on April 15, 2023 in the amount of $300,000. Jake's adjusted basis in the building was $250,000....
-
Concentration ratios have often been used to note the tightness of an oligopoly market. A high concentration ratio indicates a tight oligopoly market, and a low concentration ratio indicates a loose...
-
_____ exists when individuals in similar situations are intentionally treated differently and the different treatment is based on an individuals membership in a protected class.
-
According to Herzbergs theory, which job factor would be considered a motivator? A. Money. B. Vacation policies. C. Quarterly parties. D. Recognition of achievement through a clinical ladder.
-
Manufacturers Southern leased high-tech electronic equipment from Edison Leasing on January 1, 2013. Manufacturers Southern has the option to renew the lease at the end of two years for an additional...
-
You ve observed the following returns on Bennington Corporation s stock over the past five years: 1 6 percent, 5 percent, 1 9 percent, 1 3 percent, and 1 0 percent.a . What was the arithmetic average...
-
Consider a database that consists of the following relations. SUPPLIER(Sno, Sname) PART(Pno, Pname) PROJECT(Jno, Jname) SUPPLY(Sno, Pno, Jno) The database records information about suppliers, parts,...
-
Consider a binary liquid mixture for which the excess Gibbs energy is given by G E /RT = Ax 1 x 2 (x 1 + 2x 2 ). What is the minimum value of A for which liquid/liquid equilibrium is possible?
-
Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.20, initially at 80C and 100 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to 70C....
-
Mr. Perry is an unmarried individual with no dependent children. He reports the following information. Wages .............................................................................. $65,000...
-
It is possible to achieve a weightless condition for a limited time in an airplane by flying in a circular arc above the earth (like a rainbow). If the plane flies at $650 \mathrm{mph}$, what should...
-
A 2 in. sch 40 pipe carries a $35^{\circ} \mathrm{API}$ distillate at $50^{\circ} \mathrm{F}(\mathrm{SG}=0.85)$. The flow rate is measured by an orifice meter, which has a diameter of $1.5...
-
A 4 in. sch 80 pipe carries water from a storage tank on top of a hill to a plant at the bottom of the hill. The pipe is inclined at an angle of $20^{\circ}$ to the horizontal. An orifice meter with...
-
A tank that is vented to the atmosphere contains a liquid with a density of $0.9 \mathrm{~g} / \mathrm{cm}^{3}$. A dip tube inserted into the top of the tank extends to a point $1 \mathrm{ft}$ from...
-
The flow rate in a $1.5 \mathrm{in}$. ID line can vary from 100 to $1000 \mathrm{bbl} /$ day, and you must install an orifice meter to measure it. If you use a differential pressure (DP) cell with a...
-
The financial statements of Apple are presented in Appendix D. The company's complete annual report, including the notes to its financial statements, is available at investor.apple.com. Instructions...
-
(a) Use integration by parts to show that (b) If f and g are inverse functions and f' is continuous, prove that (c) In the case where f and t are positive functions and b > a > 0, draw a diagram to...
-
(a) What size vessel holds 2 kg water at 80C such that 70% is vapor? What are the pressure and internal energy? (b) A 1.6 m 3 vessel holds 2 kg water at 0.2 MPa. What are the quality, temperature,...
-
For water at each of the following states, determine the internal energy and enthalpy using the steam tables. (a) (b) (c) | 0 T(C) 100 550 475 180 P(MPa) 0.01 6.25 7.5 0.7
-
A permeation membrane separates an inlet air stream, F, (79 mol% N 2 , 21 mol% O 2 ), into a permeate stream, M, and a reject stream, J. The inlet stream conditions are 293 K, 0.5 MPa, and 2 mol/min;...
-
Refer to Jable 1520. Table 1521, Table 1523 and Table 1524. Two depository institutions have composite CAMELS ratings of 1 or 2 and are "well capitalized." Thus, each institution falls into the FDIC...
-
What are the financial expectations of Cann the company that produces cannabis infused beverages
-
An asset used in a four-year project falls in the five-year MACRS class for tax purposes. The asset has an acquisition cost of $5,550,000 and will be sold for $1,775,000 at the end of the project. If...

Study smarter with the SolutionInn App


