If U is considered a function of T and P, the natural heat capacity is neither C
Question:
If U is considered a function of T and P, the “natural” heat capacity is neither CV nor CP, but rather the derivative (∂U/∂T)P . Develop the following connections between (∂U/∂T)P , CP, and CV:
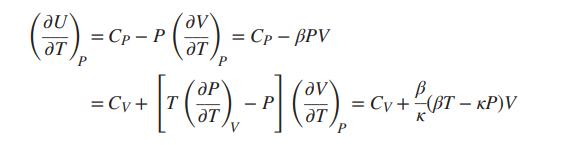
To what do these equations reduce for an ideal gas? For an incompressible liquid?
Transcribed Image Text:
(27) = C₂-P (27) = C₂ P =Cp-BPV ++ [* (*), -*] (@x), -cv+fur - t · T кP)V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The equations you provided seem to involve a mix of symbols and notation that are not standard in th...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781260597684
9th International Edition
Authors: J.M. Smith, Mark Swihart Hendrick C. Van Ness, Michael Abbott
Question Posted:
Students also viewed these Engineering questions
-
The electric and the magnetic field associated with an E.M. wave, propagating along the +z-axis, can be represented by ) [E = Eo, B = Boj] (b) [E = Eok, B = Bo] (c) [E = Eo, B = Bo] (d) [E = E), B =...
-
For a Van der Waals gas find: (a) The equation of the adiabatic curve in the variables T, V; (b) The difference of the molar heat capacities Cp = Cv as a function of T and V.
-
The specific heat at constant pressure for an ideal gas is given by cp = 0.9 + (2.7 x 10-4) T (kJ/kg K) where T is in kelvin. The change in the enthalpy for this ideal gas undergoing a process in...
-
Sumit's age after 12 years will be 6 times his age 8 years back. What is the present age of Sumit? (a) 10 (b) 12 (c) 14 (d) 15 (e) 18
-
Suppose that on the basis of the analysts past record, you estimate that the relationship between forecast and actual alpha is: Actual abnormal return = .3 Forecast of alpha Use the alphas from...
-
Distinguish between by-products and joint products. LO.1
-
What is the preemptive right of common stockholders? AppendixLO1
-
(Single-step Income, Retained Earnings, Periodic Inventory) Presented below is the trial balance of Mary J. Blige Corporation at December 31, 2004. A physical count of inventory on December 31...
-
Nature's Way Inc. is planning to invest in new manufacturing equipment to make a new garden tool. The new garden tool is expected to generate additional annual sales of 2,500 units at $60 each. The...
-
Paul and Wayne equally own the PW Partnership. Paul's basis was $30,000 and Wayne's basis was $22,000 at the beginning of the year. PW Partnership had the following income and expense items:...
-
A steam plant operates on the cycle of Fig. 8.4. The pressure levels are 10 kPa and 6000 kPa, and steam leaves the turbine as saturated vapor. The pump efficiency is 0.70, and the turbine efficiency...
-
Anglo American plc is a multinational mining company with its headquarters in South Africa. It is the worlds largest producer of platinum as well as a major producer of diamond, copper, nickel, and...
-
Charles Watts Electronics manufactures the following six peripheral devices used in computers especially designed for jet fighter planes; internal modems, external modems, graphics circuit boards,...
-
5) A frictionless rod of length L rotates counterclockwise in the with constant angular speed w at an angle a to the z axis. A bead of mass m, free to slide on the rod, leaves the origin with initial...
-
1) Louisa is a corn farmer in Illinois. She anticipates a harvest in August of 3 million bushels of yellow corn. Today is May. Louise plans to hedge her sale of corn in August using corn futures...
-
2. DETAILS MY NOTES In a statistical test, we have a choice of a left-tailed test, a right-tailed test, or a two-tailed test. Is it the null hypothesis or the alternate hypothesis that determines...
-
2. The model of a two-story building shown in Figure 2. The girders are assumed to be rigid, and the columns have flexural rigidities EI and EI2, with negligible masses. The stiffness of each column...
-
Prepare journal entries to record these transactions. (List all debit entries before credit entries. Credit account titles are automatically indented when amount is entered. Do not indent manually....
-
Refer to the situation described in BE 3-2. Determine the year-end balance in retained earnings for K and J Nursery, Inc. In BE 3-2 The trial balance for K and J Nursery, Inc., listed the following...
-
TRUE-FALSE QUESTIONS 1. In terms of preliminary analytical procedures, assume that the company has introduced a new product with a low price point and significant customer demand. The auditor would...
-
The actone(1) + chloroform(2) system has an azeotrope at x 1 = 0.38, 248 mmHg, and 35.17C. Fit the Wilson equation, and predict the P-x-y diagram.
-
Show that Wilsons equation reduces to Florys equation when A ij = A ji = 0. Further, show that it reduces to an ideal solution if the energy parameters are zero, and the molecules are the same size.
-
As part of a biorefining effort, butanediols are being produced by fermentation. The problem is that the isomers are all mixed up. Furthermore, 1,3-propanediol comprises roughly 30mol% of the mixture...
-
Construction of consumer price index number for the given goods and services. Item Weight in % Base period price Current period price Food 35 150 145 Fuel 10 25 23 Cloth 20 75 65 Rent 15 30 30 Misc....
-
Gammaro Corporation has found that 80% of its sales in any given month are credit sales, while the remainder are cash sales of the credit sales, Gammaro Corporation has experienced the following...
-
Swifty Company estimates that 2022 sales will be $43,200 in quarter 1,$51,840 in quarter 2 , and $62,640 in quarter 3 , Cost of goods sold is 50% of sales. Management desires to have ending...

Study smarter with the SolutionInn App


