Suppose a vessel contains an equimolar mixture of chloroform(1) and triethylamine(2) at 25C. The following data are
Question:
Suppose a vessel contains an equimolar mixture of chloroform(1) and triethylamine(2) at 25°C. The following data are available at 25°C: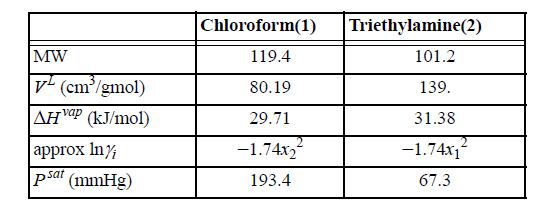
(a) If the pressure in the vessel is 90 mmHg, is the mixture a liquid, a vapor, or both liquid and vapor? Justify your answer.
(b) Provide your best estimate of the volume of the vessel under these conditions. State your assumptions.
Transcribed Image Text:
MW V(cm³/gmol) AH Vap (kJ/mol) approx In/ psat (mmHg) Chloroform(1) 119.4 80.19 29.71 -1.74x₂ 193.4 Triethylamine(2) 101.2 139. 31.38 -1.74x1 67.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
To answer these questions we will use Raoults Law and the given data Raoults Law states that the partial pressure of each component of an ideal mixtur...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
A closed system contains an equimolar mixture of n-pentane and isopentane. (a) Suppose the system is initially all liquid at 120C and a high pressure, and the pressure is gradually reduced at a...
-
Vapor?liquid equilibrium data for mixtures of acetone (A) and ethanol at 1 atm are given in the following table: (a) Use the given data to construct a Txy diagram for this system. (b) A thermocouple...
-
An equimolar mixture of n-butane and n-hexane at pressure P is brought 10 a temperature of 95C, where it exists as a vapor/liquid mixture in equilibrium. If the mole fraction of n-hexane in the...
-
Explain the Mechanism & routes of administration of SEDDS (Self emulsifying drug delivery system).
-
How does the number of firms in an oligopoly affect the outcome in its market?
-
In your opinion, does it make sense for AllRoad Parts to use FBA? Justify your answer.
-
You are invited to your six-year-old nieces birthday party and bring her the new superhero doll being advertised on television. Shes thrilled when she unwraps the gift but is in tears a short time...
-
1. What is the macro and industry environment in the Southeast Asian region for the entrance of new budget airlines? What opportunities and challenges are associated with this environment? 2. How...
-
Dure Corporation's cost formula for its selling and administrative expense is $24,500 per month plus $1 per unit. For the month of July, the company planned for activity of 7,200 units, but the...
-
A liquid mixture of 50 mol% chloroform(1) and 50% 1,4-dioxane(2) at 0.1013 MPa is metered into a flash drum through a valve. The mixture flashes into two phases inside the drum where the pressure and...
-
Using the data from problem 11.18, fit the two-parameter Margules equation, and then generate a P-x-y diagram at 78.15C. Data from problem 11.18 For the Margules two-parameter model estimate the...
-
Figure P312 shows a small truss spanning between solid supports and suspending a 10.5 kN load. The cross sections for the three main types of truss members are shown. Compute the stresses in all of...
-
The following information is available for two different types of businesses for the 2011 accounting period. Dixon Consulting is a service business that provides consulting services to small...
-
Marino Basket Company had a \(\$ 6,200\) beginning balance in its Merchandise Inventory account. The following information regarding Marino's purchases and sales of inventory during its 2011...
-
On March 6, 2011, Bob's Imports purchased merchandise from Watches Inc. with a list price of \(\$ 31,000\), terms \(2 / 10, n / 45\). On March 10, Bob's returned merchandise to Watches Inc. for...
-
The following events apply to Tops Gift Shop for 2012, its first year of operation: 1. Acquired \(\$ 45,000\) cash from the issue of common stock. 2. Issued common stock to Kayla Taylor, one of the...
-
Indicate whether each of the following costs is a product cost or a period (selling and administrative) cost. a. Transportation-in. b. Insurance on the office building. c. Office supplies. d. Costs...
-
On January 1, 2017, Wade Crimbring, Inc., a dealer in used manufacturing equipment, sold a CNC milling machine to Fletcher Bros., a new business that plans to fabricate utility trailers. To conserve...
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
(a) A pure substance is described by an expression for G(T, P). Show how to determine Z, U, and C V , in relation to G, T, and P and/or derivatives of G with respect to T and P. (b) A pure substance...
-
(a) A pure substance is described by an expression for G(T, P). Show how to determine Z, U, and C V , in relation to G, T, and P and/or derivatives of G with respect to T and P. (b) A pure substance...
-
The derivative (U / V) T is sometimes called the internal pressure and the product T(P / T) V the thermal pressure. Find equations for their evaluation for: (a) An ideal gas; (b) A van der Waals...
-
ABC Corporation has an activity - based costing system with three activity cost pools - Machining, Setting Up , and Other. The company's overhead costs, which consist of equipment depreciation and...
-
Consolidated Balance Sheets - USD ( $ ) $ in Thousands Dec. 3 1 , 2 0 2 3 Dec. 3 1 , 2 0 2 2 Current assets: Cash and cash equivalents $ 9 8 , 5 0 0 $ 6 3 , 7 6 9 Restricted cash 2 , 5 3 2 Short -...
-
How does corporate governance contribute to investor confidence and stakeholder trust? Accounting

Study smarter with the SolutionInn App


