Using the van Laar model and the data from problem 11.3, estimate the total pressure and composition
Question:
Using the van Laar model and the data from problem 11.3, estimate the total pressure and composition of the vapor in equilibrium with a 20 mol% ethanol(1) solution in water(2) at 78.15°C.
Data from problem 11.3
After fitting the two-parameter Margules equation to the data below, generate a P-x-y diagram at 78.15 °C.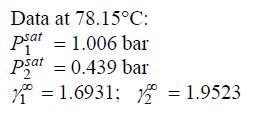
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:





