In this problem we seek to develop an expression for the van der Waals constants a and
Question:
In this problem we seek to develop an expression for the van der Waals constants a and b in terms of molecular parameters using the Sutherland model for potential energy.
(a) Show that writing the van der Waals model in virial form gives an expression for the second virial coeffi cient as: B = b - a/RT . The following mathematical relation is useful: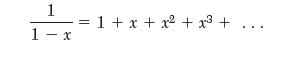
(b) Develop an expression for the second virial coeffi cient from the Sutherland model. In doing this, use the series expansion for the exponential,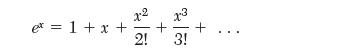
and only keep the fi rst two terms.
(c) Relate the results from parts A and B to develop an expression for the van der Waals parameters, a and b.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





