The Gibbs energy of mixing, gmix in [J/mol], of a binary liquid mixture of water (1) and
Question:
The Gibbs energy of mixing, Δgmix in [J/mol], of a binary liquid mixture of water (1) and 1-propanol (2) vs. mole fraction water (x1)is shown below. These data are at a temperature at 40°C.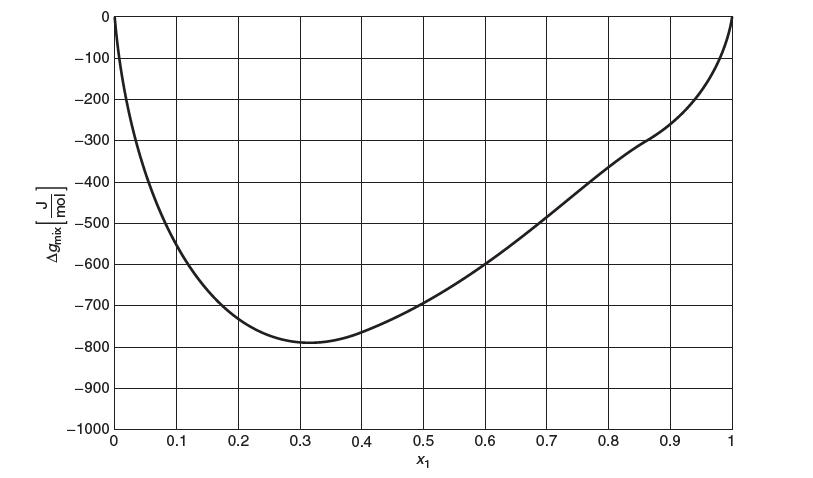

Transcribed Image Text:
A9mix mol! 0 -100 -200 -300 -400 -500 -600 -700 -800 -900 -1000 0 0.1 0.2 0.3 0.4 0.5 X1 0.6 0.7 0.8 0.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Alakhdeep Singh
I am teaching in chegg site and I am taking home tuition also
Till now I have teached above 100 students
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
How long will it take will it take a 1500 kW motor to lift a 400 kg piano to a sixt-storey window 20 m above the ground?
-
Determine the slope and displacement at point C. EI is constant. Assume A is a pin. Use the method of virtual work. A 2 k/ft 12 ft B Probs. 8-30/31 6 ft C
-
The excess Gibbs energy of solutions of methylcyclohexane (MCH) and tetrahydrofuran (THF) at 303.15 K was found to fit the expression GE=RTx (1- x) {0.4857 - 0.1077(2x -1) + 0.0191(2x- 1)2} where x...
-
Based on Exhibits 5 and 6, the value of the lower one-period forward rate is closest to: A. 3.5122%. B. 3.5400%. C. 4.8037%. Meredith Alvarez is a junior fixed-income analyst with Canzim Asset...
-
Review the case study "Alaska Airlines: Navigating Change" and then complete the following: https://www.kotterinternational.com/8-steps-process-for-leading-change/ A- State what actually occurred in...
-
Solve the given problems. The volume of a frustum of a pyramid is (This equation was discovered by the ancient Egyptians.) If the base of a statue is the frustum of a pyramid, find its volume if a =...
-
The SEC have objected to the use of the fair override on the grounds that fair is achieved by compliance with standards and that the override could lead to companies concealing poor performance and...
-
John Piderit, the general manager of the Western Tool Company, is considering introducing some new tools to the companys product line. The top management of the firm has identified three types of...
-
Question 11 (2 marks) Which TWO of the following can be included in the cost of property plant and equipment according to IAS 16? ( i ) Legal fees (ii) Staff training costs (iii) Initial testing...
-
A binary mixture of species a and b behaves as an ideal gas at 300 K and 1 bar. Calculate the partial molar Gibbs energy of species a, Ga, and the total solution Gibbs energy, g, at the following...
-
A group of students came across an unsuspected supply of laboratory alcohol containing 96 weight % ethanol (EtOH) and 4 weight % water (H2O) . As an experiment, they wanted to see ifthey could make...
-
Identify positive and negative emotions and describe how emotions affect behavior.
-
what is wrong with this code? identifier errors and fix it import java.util.Scanner; public class HandsOn { public static void main(String[] args){ int val = 0; int count = 0; Scanner in = new...
-
What is the largest and smallest complete (all major organelles)cells in the human body? Is it smallest- cell and biggest -organism? or should I go even more back and say that element, atom as...
-
Two alternatives, identified X and Y, are under consideration at Hayden Corporation. Costs associated with the alternatives are listed below. Material costs Processing costs Building costs Equipment...
-
JCBilco Manufacturing produces and sells oil filters for $3.35 each. A retailer has offered to purchase 10,000 oil filters for $3.30 per filter. Of the total manufacturing cost per filter of $2.15,...
-
Suppose a home is purchased for $325,000 and then sold 6 months later for $400,000 after spending $25,000 for capital improvements on the home. The homeowner's taxable income for the year is $160,000...
-
Explain any possible differences between accounting for an account receivable factored with recourse compared with one factored without recourse.
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
Discuss the physical origins of the gross selection rules for rotational and vibrational Raman spectroscopy.
-
Suppose that you wish to characterize the normal modes of benzene in the gas phase. Why is it important to obtain both infrared absorption and Raman spectra of your sample?
-
Calculate the ratio of the Einstein coefficients of spontaneous and stimulated emission, A and B, for transitions with the following characteristics: (a) 500 MHz radiofrequency radiation, (b) 3.0 cm...
-
Net Present Value A project has estimated annual net cash flows of $10,000 for ten years and is estimated to cost $42,500. Assume a minimum acceptable rate of return of 20%. Use the Present Value of...
-
Shrey ASSIGNMENT 2) Each of the program budgets must be developed using the information below and using object classification numbers provided 1) This is a time-consuming assignment In your note...
-
Which of the following provisions of the CARES Act does California law conform to? a. Taxpayers were temporarily exempt from the early withdrawal penalty from 401(k) plans. b. There is a five-year...

Study smarter with the SolutionInn App


