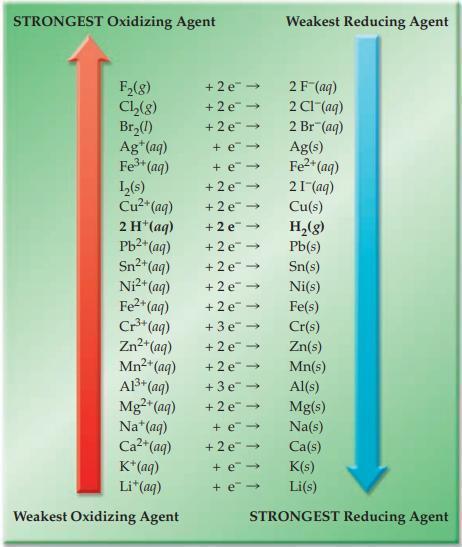
Refer to Figure 17.4 and state whether each of the following ionic redox reactions is spontaneous or
Question:
Refer to Figure 17.4 and state whether each of the following ionic redox reactions is spontaneous or nonspontaneous.
(a) Mg(s) + Sn2+(aq) → Mg2+(aq) + Sn(s)
(b) H+(aq) + Ni(s) → H2(g) + Ni2+(aq)
Figure 17.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:





