Silicon is the second most abundant element in Earths crust. Calculate the atomic mass of silicon given
Question:
Silicon is the second most abundant element in Earth’s crust. Calculate the atomic mass of silicon given the following data for its three natural isotopes:
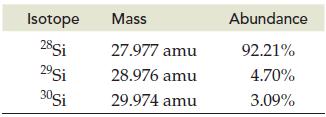
Transcribed Image Text:
Isotope 28Si 29Si 30Si Mass 27.977 amu 28.976 amu 29.974 amu Abundance 92.21% 4.70% 3.09%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
We can find the atomic mass of silicon as follows The av...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The thin outer layer of Earth, called the crust, contains only 0.50 percent of Earth's total mass and yet is the source of almost all the elements (the atmosphere provides elements such as oxygen,...
-
Although He is the second most abundant element in the universe, it is very rare on the earth. Why?
-
Gerta is 30 years old and advises her financial planner that she wants to retire in 35 years. The financial planner and Gerta determine that Gerta will need to receive beginning of the month payments...
-
Use f(x) and g(x) to find a formula for each expression. Identify its domain. (a) (f + g)(x) (c) (fg)(x) (b) (f- g)(x) (d) (f/g)(x)
-
Factors Affecting Financial Condition. The transmittal letter from the chief financial officer to the mayor and city council of Detroit that accompanies the City of Detroits Comprehensive Annual...
-
Quinton Johnston is evaluating TMI Manufacturing Company, Ltd., which is headquartered in Taiwan. In 2008, when Johnston is performing his analysis, the company is unprofitable. Furthermore, TMI pays...
-
The average age of the 115 residents of a retirement community
-
Sutherland manufactures and sells 60,000 laser printers each month. A principal component part in each printer is its paper feed drive. Sutherland's plant currently has the monthly capacity to...
-
The financial statements of PSG are presented in Appendix B. The company's complete annual report, including the notes to the financial statements, is available online. Click here to view Appendix B....
-
Which subatomic particle has a relative charge of +1 and a mass of 1 amu? (a) Alpha (b) Electron (c) Neutron (d) Proton (e) None of the above.
-
State three scientists whose work Dalton used to support the atomic theory.
-
Tandin is a director and officer of United Products, Inc. Tandin makes a decision about the marketing of Uniteds products that results in a dramatic decrease in profits for United and its...
-
A farmer has an acre of specialty vegetables and is preparing for the summer harvest. Historically, this acre has yielded an average of 2,100 lbs of product with a standard deviation of 950 lbs. A...
-
Solve 3x 82+22 = (4).
-
(c) Compute EVPI and EVSI (in thousands of dollars). (Round your answers to one decimal place.) EVPI $ 3.6 EVSI $ 3.6 Xthousand x thousand Discuss whether the firm should consider a consulting expert...
-
Question 9 (1 point) If the common law requires employees of a bar establishment to monitor a potentially intoxicated patron and to possibly make an effort to intervene if there is an indication the...
-
B. A velocity potential is given by the equation: Q = x-y 3. (10 pts) Short answer, What special characteristics of the velocity potential make it very useful in identifying a type of flow and...
-
Rank the following compounds in order of decreasing stability: trans-3-hexene, cis-3-hexene, 1-hexene, cis-2,2,5,5-tetramethyl-3-hexene
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
How does accounting help the capital allocation process?
-
What are some of the major challenges facing the accounting profession?
-
What are the major objectives of financial reporting?
-
What is Coke's average ownership percentage in its equity method investments? Goodwill is 7000 Calculate the firm's current ratio (current assets/current liabilities). Calculate the current ratio...
-
John has to choose between Project A and Project B, which are mutually exclusive. Project A has an initial cost of $30,000 and an internal rate of return of 16 percent. Project B has an initial cost...
-
Complete the table below, for the above transactions

Study smarter with the SolutionInn App


