Crystallographers and materials scientists use the density of a metallic sample to infer its likely crystal structure.
Question:
Crystallographers and materials scientists use the density of a metallic sample to infer its likely crystal structure. The density of copper (Cu) is 8.96 g/cm3 and its atomic radius is 0.128 nm. Is the copper crystal more likely to be face-centered cubic or body-centered cubic? (See Figure 10-57.)
Figure 10-57
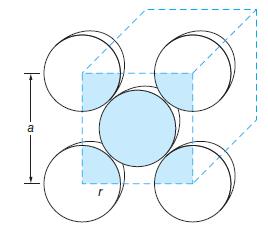
Transcribed Image Text:
a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Crystallographers and materials scientists often use the relationship between the density and the cr...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The density of a sample of HCP beryllium is 1.844 g/cm3, and the lattice parameters are 0 = 0.22858 nm and c0 = 0.35842 nm. Calculate (a) The fraction of the lattice points that contain vacancies;...
-
The density of a sample of FCC palladium is 11.98 g/cm3, and its lattice parameter is 3.8902 . Calculate (a) The fraction of the lattice points that contain vacancies; and (b) The total number of...
-
Lakefield Corporations oil trading desk buys and sells oil products (crude oil and refined fuels), options, and futures in international markets. The trading desk is responsible for buying raw...
-
During a worldwide recession in 1983, the oil cartel began to lower prices. Why would a recession make the cartel more vulnerable to price cutting? How would the reduced demand be shared between the...
-
The following transactions apply to Midsouth Equipment Sales Corp. for 2013: 1. The business was started when Midsouth received $60,000 from the issue of common stock. 2. Purchased $160,000 of...
-
In the parallel network of Figure 11.9 there is no current flowing in either loop prior to closing the switch at time t = 0. Deduce the currents i 1 (t) and i 2 (t) flowing in the loops at time t....
-
1. DiversCo, a large U.S. company, operates in two areas: energy and retail clothing. You observe an analyst report that valuesDiversCo using multiples analysis based on a peer group of other large...
-
On August 1, Rantoul Stores Inc. is considering leasing a building and purchasing the necessary equipment to operate a retail store. Alternatively, the company could use the funds to invest in...
-
here is all the info for the exercise. Residual Plots for Y Normal Probability Plot Versus Fits File Histogram Versus Order 21434 Curwone Resid I WORSHEET Probability Plot of RESI Probability Plot of...
-
In the anomalous Zeeman effect, the external magnetic field is much weaker than the internal field seen by the electron as a result of its orbital motion. In the vector model the vectors L and S...
-
Approximating atoms in an FCC crystal as hard spheres of radius r with a being the length of each side of the unit cube, what fraction of the volume of the cube (and hence the crystal) is occupied by...
-
Describe the effect of a distribution in a year when the distributing corporation has: a. A deficit in accumulated E & P and a positive amount in current E & P. b. A positive amount in accumulated E...
-
Let $N$ be a positive integer. Consider the relation $\circledast$ among pairs of integers $r, s \in \mathbb{Z}$ defined as $r \circledast s$ when $r-s$ is an integer multiple of $N$. Prove that...
-
Draw a circuit diagram for a typical home hair dryer. To which form (or forms) of energy is electric potential energy converted when you use the dryer?
-
Draw a vector field diagram for particles carrying charges \(+2 q\) and \(-q\) separated by a distance \(r\). Comment on the significance of the vector diagram.
-
(a) Show that the Jones matrix of a polarization analyzer set at angle \(\alpha\) to the \(X\)-axis is given by \[ \underline{\mathbf{L}}(\alpha)=\left[\begin{array}{cc} \cos ^{2} \alpha & \sin...
-
Let \(\mathbf{V}(t)\) be a linearly filtered complex-valued, wide-sense stationary random process with sample functions given by \[ \mathbf{v}(t)=\int_{-\infty}^{\infty} \mathbf{h}(t-\tau)...
-
According to research, have mutual funds outperformed the market? Explain. Would mutual funds be attractive to some investors even if they are not expected to outperform the market? Explain.
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
A rocket-powered hockey puck has a thrust of 2.0 N and a total mass of 1.0 kg. It is released from rest on a frictionless table, 4.0 m from the edge of a 2.0 m drop. The front of the rocket is...
-
The father of Example 8.2 stands at the summit of a conical hill as he spins his 20 kg child around on a 5.0 kg cart with a 2.0-m-long rope. The sides of the hill are inclined at 20. He again keeps...
-
The father of Example 8.2 stands at the summit of a conical hill as he spins his 20 kg child around on a 5.0 kg cart with a 2.0-m-long rope. The sides of the hill are inclined at 20. He again keeps...
-
7 . 4 3 Buy - side vs . sell - side analysts' earnings forecasts. Refer to the Financial Analysts Journal ( July / August 2 0 0 8 ) study of earnings forecasts of buy - side and sell - side analysts,...
-
Bond P is a premium bond with a coupon of 8.6 percent , a YTM of 7.35 percent, and 15 years to maturity. Bond D is a discount bond with a coupon of 8.6 percent, a YTM of 10.35 percent, and also 15...
-
QUESTION 2 (25 MARKS) The draft financial statements of Sirius Bhd, Vega Bhd, Rigel Bhd and Capella for the year ended 31 December 2018 are as follows: Statement of Profit or Loss for the year ended...

Study smarter with the SolutionInn App


