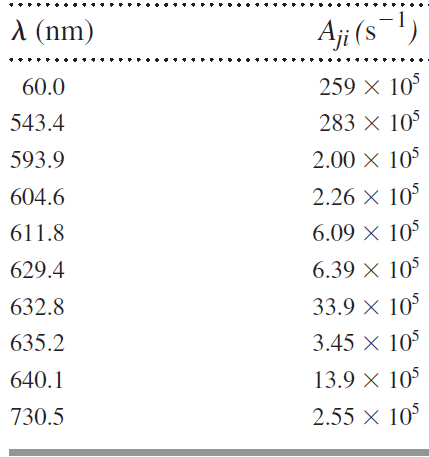
The helium-neon laser is famous for its red-light emission at 632.8 nm. But electrons in that same
Question:
Table 13.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





