Cinnamaldehyde is one of the primary constituents of cinnamon oil and contributes significantly to the odor of
Question:
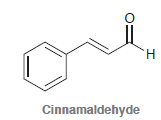
н Cinnamaldehyde
Step by Step Answer:
H N...View the full answer



Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Sciences questions
-
Limonene is a hydrocarbon found in the peels of lemons and contributes significantly to the smell of lemons. Limonene has a molecular ion peak at m/z = 136 in its mass spectrum, and it has two double...
-
Cinnamaldehyde is used as a flavoring agent in cinnamon candies. Show how cinnamaldehyde is synthesized by a crossed aldol condensation followed by dehydration. cinnamaldehyde-C
-
Starting with benzene and using any other necessary reagents of your choice, design a synthesis for the following compound. -NH2 -
-
The Sales Discounts account is a contra account to which of the following accounts? Cost of Goods Sold Sales Returns and Allowances Purchases Discounts Sales Revenue
-
Is police work a profession? Explain. What are the advantages of viewing policing as a profession? How can police professionalism be enhanced?
-
Consider the figure. The area of the entire colored region is represented by__________ , because each side of the entire colored region has length _______. a b b
-
Causes of death. Government data assign a single cause for each death that occurs in the United States. The data show that the probability is 0.27 that a randomly chosen death was due to heart...
-
Suppose there's an appliance store that sells refrigerators. It could set its price high and sell very few refrigerators, or it could set its price low and sell many more refrigerators. The following...
-
Consider the APT. The risk premiums on factor 1 and factor 2 portfolios are 5% and 3%, respectively. The risk-free rate of return is 1.5%. Stock A has an expected return of 19% and a beta on factor 1...
-
Exercise 2.9 Use the planar version of Grbler's formula to determine the number of degrees of freedom of the mechanisms shown in Figure 2.18. Comment on whether your results agree with your intuition...
-
The American Cancer Society is beginning a study to learn why some people never get cancer. To take part in the study, a person must be 3065 years of age and never had cancer. The study requires that...
-
Rats with cancer are divided into two groups. One group receives 5 milligrams (mg) of a medication that is thought to fight cancer, and the other receives 10 mg. After 2 years, the spread of the...
-
Think of a job that you have held or that was held by someone you know. If you were going to analyze that job using one of the methods in the book, which method would you use and why?
-
I need help with discussion posts that respond to 3 of these comments. 2 of them being the first on each picture. RUBRIC: articles to mention Coleman, R., & Banning, S. (2006). Network TV news'...
-
2. Best Use of Scarce Resource DigiCom Corporation produces three sizes of television sets: 12-inch screen, 26-inch screen, and 40-inch screen. Revenue and cost information per unit for each product...
-
Gunther invested $15,000 into a segregated fund with a 65% maturity guarantee 10 years ago. The fund is now maturing and has a current market value of $22,261. Gunther decides to withdraw his...
-
(a) Consider the following financial data (in millions of dollars) for Costello Laboratories over the period of 2014-2018: Year Sales Net income Total assets Common equity 2014 $3,800 $500 $3,900...
-
The Pizza Pie 'N Go sells about 2300 one-topping pizzas each month. The circle graph displays the most requested one-topping pizzas, by percentage, for one month. Most Popular One-Topping Pizzas...
-
Prove the sum rule for derivatives. Note that the difference quotient for f + g can be written as (f+ g)(x + h) (f + g)(x) h = [f(x + h) + g(x + h)] - [f(x) + g(x)] h
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
Outline a preparation of each of the following from an amine and an acid chloride. (a) N-phenylbenzamide (b) N-benzyl-N-ethylpropanamide
-
What product (including its stereochemistry) is expected from the Hofmann elimination of each of the following stereoisomers? N(CH3)3 OH (2R,3S)- Ph CH CH Ph CHy
-
Give an acceptable name for the following compounds. (a) (b) CH3 CH3 NH
-
Assignment Title: The Role of Bookkeeping in Business Management and Financial Reporting Objective: Understand the importance of proper bookkeeping procedures in the management of...
-
17) The adjustment that is made to allocate the cost of a building over its expected life is called:A) depreciation expense.B) residual value.C) accumulated depreciation.D) None of the above answers...
-
9) Prepaid Rent is considered to be a(n):A) liability.B) asset.C) contra-asset.D) expense.10) As Prepaid Rent is used, it becomes a(n):A) liability.B) expense. C) contra-asset.D) contra-revenue.11)...

Study smarter with the SolutionInn App


