Draw the expected 1 H NMR spectrum of the following compound: CI
Question:
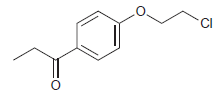
Transcribed Image Text:
CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
22 8 7 ...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the expected 1 H NMR spectrum for each of the following compounds: (a) (b) (c) H.
-
Draw the expected NMR spectrum of methyl propionate, and point out how it differs from the spectrum of ethyl acetate. CH--0-C-CH,--CH, methyl propionate
-
Draw the integral trace expected for the NMR spectrum of tert-butyl acetoacetate, shown in Figure 13-17. CH3 C-O-CCH3 3 0 6 10 (ppm)
-
Following are the most recent balance sheets for Country Kettles, Inc. Excluding accumulated depreciation, determine whether each item is a source or a use of cash, and the amount. COUNTRY KETTLES,...
-
What are three examples of best practices in B2B e-commerce environment?
-
The cross-tabulation data given below represent the number of males and females in a survey who have or have not visited an urgent care facility in the last month. a. Write the hypotheses for the...
-
For a hypothetical ethical dilemma you develop, use the eight-stage process for making a sound ethical decision. AppendixLO1
-
Liane Hansen has prepared the following list of statements about bonds. 1. Bonds are a form of interest-bearing notes payable. 2. When seeking long-term financing, an advantage of issuing bonds over...
-
0 Required information [The following information applies to the questions displayed below) Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company...
-
In the potential outcomes framework with heterogeneous (nonconstant) treatment effect, write the error as u i = (1 x i )u i (0) + x i u i (1). Let s 2 0 = Var[u i (0)] and s 2 1 Var[ui(1)]. Assume...
-
The city of Gainesville issued $1,000,000 of 14 percent coupon, 30-year, semiannual payment, tax-exempt muni bonds 10 years ago. The bonds had 10 years of call protection, but now Gainesville can...
-
The Hawley Corporation is attempting to determine the optimal level of current assets for the coming year. Management expects sales to increase to approximately $2 million as a result of an asset...
-
In Problems 1956, factor completely. If a polynomial cannot be factored, say so. x 2 - 4xy - 12y 2
-
The adjusted trial balance section of Menlo Company's worksheet shows a \(\$ 1,500\) debit balance in utility expense. At the end of the accounting period the accounting manager accrues an additional...
-
Identify each of the 10 amount columns of the worksheet and indicate to which column the adjusted balance of the following accounts would be extended: a. Accounts Receivable b. Accumulated...
-
Using the data from Table 3.3, show the effect on world output if each country moved toward specialization in the production of its comparative-disadvantage good. TABLE 3.3 Comparative Advantage as a...
-
The Professional Winner was RJ Andrews from Info We Trust, for the video Are Gazelles Endangered? (a) Watch this video. What data are this video conveying? (b) You can interact with the data and...
-
(a) Draw a simplified ray diagram showing the three principal rays for an object located outside the focal length of a converging lens. (b) Is the image real or virtual? (c) Is it upright or...
-
According to one model, the future increases in average global temperatures (due to car- bon dioxide levels exceeding 280 parts per million) can be estimated using T = 6.5 In (C/280), where C is the...
-
Record the following selected transactions for March in a two-column journal, identifying each entry by letter: (a) Received $10,000 from Shirley Knowles, owner. (b) Purchased equipment for $35,000,...
-
Order the compounds in each of the following sets with respect to increasing acidity: (a) Acetic acid, oxalic acid, formic acid (b) p-Bromobenzoic acid, p-nitro benzoic acid, 2, 4-dinitrobenzoic acid...
-
Arrange the compounds in each of the following sets in order of increasing basicity: (a) Magnesium acetate, magnesium hydroxide, methyl magnesium bromide (b) Sodium benzoate, sodium p-nitro benzoate,...
-
How could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1 -Butanol (b) 1Bromobutane (c) Pentanoic acid (d) 1-Butenc (e) Octane
-
Ted and his partners have contracted to purchase the franchise nights worth 561 000 to open and operate a specialty pizza restaurant called Popper with a renewable agrement, the partners have agreed...
-
Your answer is partially correct. Martin Company's chief financial officer feels that it is important to have data for the entire quarter especially since their financial forecasts indicate some...
-
Kellog Corporation is considering a capital budgeting project that would have a useful life of 4 years and would love testing 5156.000 in equipment that would have zeto salvage value at the end of...

Study smarter with the SolutionInn App


