Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each
Question:
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required.
a.
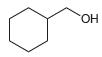
b.
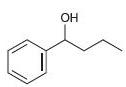
c.
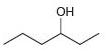
d.

Transcribed Image Text:
HO. HO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a ...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how each of the following alcohols can be made from a Grignard reagent and a carbonyl compound: a. b. 0 0
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Assume a portfolio of two assets, with $10m invested in asset 1 and $5m invested in asset 2. The per pound covariance matrix of the two assets is: Asset 1 Asset 2 The best hedge of asset 2 is: Asset...
-
Kessler describes one of the goals of religious studies as "making the strange familiar" and "making the familiar strange" (xiii). What do you think he means by that? Can you think of any specific...
-
The unadjusted and adjusted trial balances for Tinker Corporation on December 31, 2015, are shown below: Journalize the nine adjusting entries that the company made on December 31, 2015.
-
5 You are a brand manager for a sneaker manufacturer like Nike or New Balance and you are trying to use Facebook to reach (a) college-aged women and (b) men over 55 years of age. What three or four...
-
Supermart Food Stores (SFS) has experienced net operating losses in its frozen food products line in the last few periods. Management believes that the store can improve its profitability if SFS...
-
Required information [ The following information applies to the questions displayed below. ] Delicious Dave's Maple Syrup, a Vermont Corporation, has property in the following states:
-
Bombay Fast Food and 2 Bros. Pizza are pizza parlors that are located a few feet away from each other on a street in New York. a. Both firms sell pizza slices at a price of $1 each. Given this price,...
-
Is the ratio of fugacity to pressure greater to or less than one if the attractive part of the interaction potential between gas molecules dominates?
-
A 1.25 mole sample of an ideal gas is expanded from 320. K and an initial pressure of 3.10 bar to a final pressure of 1.00 bar, and C P,m = 5/2R. Calculate w for the following two cases: a. The...
-
What properties are essential in an equation that may be solved independently of the remaining equations in a system of equations?
-
Part 2 Problems 1. Jets Corp. maintains its books on a cash basis. However, the company obtained a loan of $150,000 from a local bank. The bank requires Jets Corp. to provide annual financial...
-
Company A has a well-developed brand website. They send e-mails to customers and prospects. However, the company has never used social media or mobility marketing. You have been called upon to...
-
Is Time Running Out for Bed Bath & Beyond case study and answer following questions: 3-13 analyze bed bath & beyond using the competitive forces and value chain models. 3-14 define the problem faced...
-
Marcus expresses an interest in learning more about Katie's job position, telling her that he hopes to be in the position himself one day. Katie decides to take Marcus under her wing and teach him...
-
Losing to a Weaker Foe What began as a heavily conventional military campaign to unseat the regime of Saddam Hussein had become a bitter, unconventional struggle against frustrated Sunnis who...
-
In Problems 18, find the real solution(s), if any, of each equation. 2x + 1 = 3
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
Show the structures of the following amino acids in their zwitterionic forms: (a) Trp (b) IIe (c) Cys (d) His
-
Proline has pKa1 = 1.99 and pKa2 = 10.60. Use the HendersonHasselbalch equation to calculate the ratio of protonated and neutral forms at pH = 2.50. Calculate the ratio of neutral and deprotonated...
-
Using both three and one-letter codes for amino acids, write the structures of all possible peptides containing the following amino acids: (a) Vat, Ser, Leu (b) Ser, Leu2, Pro
-
Series of Compound Interest Techniques The following are several situations involving compound interest. Required: Using the appropriate table, solve each of the following: ( Click here to access the...
-
If Clark Kelly has recognized gain on an exchange of like-kind property held for investment use, where does Clark report the gain? First on Form 8824, then carried to Schedule D. First on Form 8824,...
-
An investor put 40% of her money in Stock A and 60% in Stock B. Stock A has a beta of 1.2 and Stock B has a beta of 1.6. If the risk-free rate is 5% and the expected return on the market is 12%,...

Study smarter with the SolutionInn App


