For each of the following pairs of compounds determine whether the two compounds would be at equilibrium
Question:
For each of the following pairs of compounds determine whether the two compounds would be at equilibrium at room temperature or whether they could be isolated from one another:
(a)
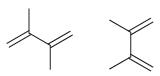
(b)
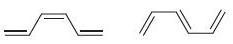
(c)
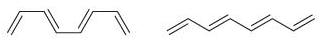
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a These drawings represent two different conformations of the same compound the scis conformation ...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, determine whether they are enantiomers, epimers, diastereomers that are not epimers, or identical compounds: (a) (b) (c) (d) . O: - - - - - - - -...
-
For each of the following pairs of compounds, determine whether they are enantiomers, epimers, diastereomers that are not epimers, or identical compounds: (a) d-Glucose and d-gulose (b)...
-
For each of the following pairs of compounds, determine which compound is more stable (you may find it helpful to draw out the chair conformations): (a) (b) (c) (d) II
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
A sub-rounded to sub-angular sand has a D10 of about 0.1 mm and a uniformity coefficient of 3. The angle of shearing resistance measured in the direct shear test was 47. Is this reasonable? Why or...
-
Table below represents the operation of an Open-Access fishery. Total Catch Total Average Marginal # of Boats (tons) Revenue Revenue Revnue 1 15 1950 1950 1950 2 30 3900 1950 1950 3 45 5850 1950 1950...
-
15-7. Promotional programs can be directed to __________, ___________, or both.
-
What conditions suggest that a ratio variable should be transformed (recoded) into a dichotomous (two group) variable?
-
Oct. Sales Rev.- 46,000 Nov.- 54,000 Dec.-39000 Sells whistle for $10. Production of 5% Question Help Whistle Works manufacturers safety whistle keychains. They have the following information...
-
Consider the structure of lysergic acid diethylamide (LSD), a potent hallucinogen containing three nitrogen atoms. One of these three nitrogen atoms is significantly more basic than the other two....
-
Diamond Hill Jewelers is considering the following independent projects: Which project(s) should be accepted if the required rate of return for the projects is 10 percent? Compute the NPV and the IRR...
-
What is the principle of competitive exclusion? Of resource partitioning?
-
At a given point on the surface of the wing of the airplane in Prob. 3.6, the flow velocity is 130 m/s. Calculate the pressure coefficient at this point.
-
1. Why do companies that choose to open subsidiaries in other countries have different HR responsibilities? 2. How has globalization allowed companies to become "global companies" more easily? 3....
-
Is Kroger's innovation Product-related or process-related? Do the innovations tend to be incremental or radical? https://www.thekrogerco.com/about-kroger/our-business/ Kroger Co. opens new spoke in...
-
Define what is Process Mapping/Value Stream Mapping How do you apply process mapping methodology? What are the advantages of leaders using process mapping Identify a real world business...
-
What role do formalized processes and protocols play in highly structured organizations, and how can organizations balance the need for structure with the imperative for flexibility and innovation ?
-
In what ways do decision-makers balance quantitative data with qualitative insights to optimize complex strategic choices, especially in high-stakes business environments where traditional metrics...
-
Problems 2837 are based on material learned earlier in the course. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for the final exam. Write...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Outline methods for the preparation of acetophenone (phenyl methyl ketone) starting from the following: (a) Benzene (b) Bromobenzene (c) Methyl benzoate (d) Benzonitrile (e) Styrene
-
The following reactivity order has been found for the basic hydrolysis of p-substituted methyl benzoates: Y = NO 2 > Br > H > CH 3 > OCH 3 . How can you explain this reactivity order? Where would you...
-
The following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH. Explain. CH3CO2CH3 > CH3CO2CH2CH3 > CH3CO2CH (CH3)2 > CH3CO2C (CH3)3
-
(15 points) Stressed $2.500,000 of S% 20 year bands. These bonds were issued Jary 1, 2017 and pay interest annually on each January 1. The bonds yield 3% and was issued at $325 8S! Required (2)...
-
Packaging Solutions Corporation manufactures and sells a wide variety of packaging products. Performance reports are prepared monthly for each department. The planning budget and flexible budget for...
-
1. A company issued 10%, 10-year bonds with a par value of $1,000,000 on January 1, at a selling price of $885,295 when the annual market interest rate was 12%. The company uses the effective...

Study smarter with the SolutionInn App


