Hydroxide is not a suitable base for deprotonating acetylene: Explain why not. Can you propose a base
Question:
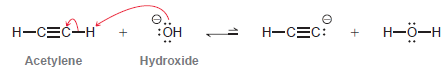
Explain why not. Can you propose a base that would be suitable?
Transcribed Image Text:
Н—СЕС-Н Н-СЕС: :Он Н-ӧ—н Acetylene Hydroxide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
The equilibrium does not favor deprotonation of acetylene by hydroxi...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Can the function = x2 (L - 2x) be used as a trial wave function for the n = 1 state of a particle with mass m in a one-dimensional box of length L? If the answer is yes, then express the energy of...
-
Time-Dependent Wave Function for a Free Particle One example of a time-dependent wave function is that of a free particle [one for which U(x) = 0 for all x] of energy E and x-component of momentum p....
-
(a) Guanidine (shown) is about as strong a base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines. (b) Show why p-nitroaniline is a much weaker base (3 pKb units...
-
Judy Baker is a new employee of Farnsdel and Babcock, LLP. Which of the following will provide proof of employment authorization for the completion of the I-9? 1. U.S. passport 2. U.S. military...
-
North Sea Oil has compiled the following data relative to current costs of its basic sources of external capitals, long-term debt, preferred stock, and common stock equity. Source of capital Cost...
-
Use Lagrange multipliers to find any extrema of the function subject to the constraint x + y 1. (x, y) = e -xy/4
-
Select an SRS. A student at a large university wants to study the responses that students receive when calling an academic department for information. She selects an SRS of 6 departments from the...
-
The year-end balance sheet of Manor, Inc., includes the following stockholders equity section (with certain details omitted): Stockholders equity: 10% cumulative preferred stock, $100 par value,...
-
\ table [ [ , Date,Units,Unit Cost,Total Cost ] , [ Beginning Inventory,January 1 , 1 2 0 , $ 8 0 , $ 9 , 6 0 0
-
This diagram shows an example of memory configuration under dynamic partitioning, after a number of placement and swapping-out operations have been carried out. Addresses go from left to right; gray...
-
Determine the position of equilibrium for each acid-base reaction below: (a) (b) (c) (d) `H. O + H20
-
Amino acids, such as glycine, are the key building blocks of proteins and will be discussed in greater detail in Chapter 25. At the pH of the stomach, glycine exists predominantly in aprotonated form...
-
A 13.6-kg block is supported by the spring arrangement shown. If the block is moved from its equilibrium 44 mm vertically downward and released, determine (a) The period and frequency of the...
-
Swenson Company produced 300 units in year one and sold 260 units in that year. In year two, it produced 260 units and sold 300 units. Total fixed overhead was the same in years one and two. Under...
-
c) Determine the maximum rotational speed such that the fluid will not spill over the container. (and: = 2gh/R) [2 marks] d) The container in Figure 4 now contains coffee (p~1000) which is 7cm deep...
-
FICO credit scores: x = 564,= 743,= 72 (Round your answer to 3 decimal places.) what does z equal
-
Q3: In the section illustrated in Figure (1) the surface 1-4-7 is insulated. The convection heat transfer coefficient at surface 1-2-3 is 28 W/m. 'C. The thermal conductivity of the solid material is...
-
25 of 27 > This test: 96 point(s) possible This question: 3 point(s) possible Submit test Identical twins come from a single egg that split into two embryos, and fraternal twins are from separate...
-
Solve each system. 4x + 2y 32 = 6 x - 4y + z 2 -x + = -4 2z = 2
-
Economic feasibility is an important guideline in designing cost accounting systems. Do you agree? Explain.
-
How would the molecular ion peaks in the respective mass spectra of CH3Cl, CH2 Cl2, CHCl3, and CCl4 differ on the basis of the number of chlorines? (Remember that chlorine has isotopes 35Cl and 37Cl...
-
Provide the reagents necessary for the following synthetic transformations. More than one step may be required. (a) (b) (c) (d) (e) (f) OCH3 Br
-
Synthesize each of the following compounds by routes that involve allylic bromination by NBS. Use starting materials having four carbons or fewer. Begin by writing a retrosynthetic analysis. (a) (b)...
-
Los datos de la columna C tienen caracteres no imprimibles antes y despus de los datos contenidos en cada celda. En la celda G2, ingrese una frmula para eliminar cualquier carcter no imprimible de la...
-
Explain impacts of changing FIFO method to weighted average method in inventory cost valuations? Explain impacts of changing Weighted average method to FIFO method in inventory cost valuations?...
-
A perpetuity makes payments starting five years from today. The first payment is 1000 and each payment thereafter increases by k (in %) (which is less than the effective annual interest rate) per...

Study smarter with the SolutionInn App


