Identify the missing reagent needed to achieve the following transformations: a. b. c. d. Br KF benzene
Question:
a.
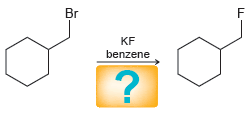
b.
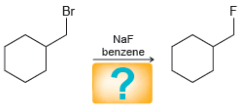
c.
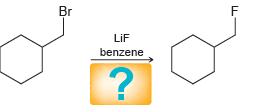
d.
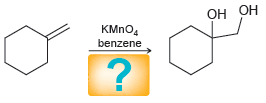
Transcribed Image Text:
Br KF benzene Br NaF benzene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b c ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide the reagents necessary to achieve the following transformations. (a) (b) (D = deuterium)
-
Show how each of the following transformations could be accomplished: (a) (b) (c) (d) (e)
-
Fill in the missing reagent(s) needed to convert each molecule into the next one pictured in the synthetic scheme below. If a transformation requires more than one step, number the reagents for the...
-
Lancaster Lumber buys $8 million of materials(net of discounts) on terms of 3/5, net 55, and it currently pays on the 5th day and takes discounts. Lancaster plans to expand, which will require...
-
Conservatives have long accused traditional media outlets of having a liberal bias. Are they correct? If so, to what degree? Regardless of whether this particular accusation is correct, whether this...
-
One cubic meter of an ideal gas at 600 K and 1,000 kPa expands to five times its initial volume as follows; (a) By a mechanically reversible, isothermal process. (b) By a mechanically reversible,...
-
Prepare the operating budget, the financial budget, and the supporting schedules.
-
The following monthly data are taken from Nunez Company at July 31: Sales salaries, $120,000; Office salaries, $60,000; Federal income taxes withheld, $45,000; State income taxes withheld, $10,000;...
-
The Grants have provided you some additional information pertaining to a construction business Bob started. This business, GPE, is a sole proprietorship with Bob being the sole owner. GPE is located...
-
a. What should the weekly product mix consist of, and what is the weekly net profit? b. Is the optimal solution to question 1 degenerate? Explain your response. c. Is the optimal solution from...
-
Equation (1.19) states that the total pressure in a mixture of gases is equal to the sum of the partial pressures. Is this equation valid for real gases? If so, under what conditions?
-
Consider the oxidation of the amino acid glycine NH 2 CH 2 COOH to produce water, carbon dioxide, and urea NH 2 CONH 2 : NH 2 CH 2 COOH(s) + 3O 2 (g) NH 2 CONH 2 (s) + 3CO 2 (g) + 3H 2 O(l)...
-
Cite the primary differences between addition and condensation polymerization techniques
-
Skippy wants to have $18,000.00 in 7 years. His bank is offering an account that earns 2% compounded monthly. How much does he need to deposit to reach his goal? Round your final answer up to the...
-
2. Three students are chosen randomly to be class president, vice president, and treasurer. No student can hold more than one office. (a) What is the probability that Mary is president, Cory is vice...
-
The current price of a non - dividend paying stock is $ 5 0 . Consider an American put option on the stock with a strike price of $ 4 8 that expires in 1 2 months. In each of the next six month...
-
Cinder Inc. is a Canadian - controlled private corporation based in your province. The company operates a wholesale business. The following information is provided for its year ended May 3 1 , 2 0 2...
-
Use part one of the fundamental theorem of calculus to find the derivative of the function. y' = 3x + 5 t y = dt 1+ 3
-
In their study of children in play therapy (see Guided Practice 9.1), Reyes and Asbrand (2005) also measured the level of depression that the children had before and after therapy. They found that D...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
What amino acid sequence is coded by the following mRNA base sequence? CUU-AUG-GCU-UGG-CCC-UAA
-
What is the base sequence in the original DNA strand on which the mRNA sequence in Problem 28.9 was made? CUU-AUG-GCU-UGG-CCC-UAA
-
p-Dimethoxytrityl (DMT) ethers are easily cleaved by mild acid treatment. Show the mechanism of the cleavage reaction.
-
Algunas acciones a desarrollar en las entregas certificadas son: Puede seleccionar ms de una opcin. Grupo de opciones de respuesta Reducir manipulacin excesiva Despacho Transporte Comunicaciones de...
-
PRACTICE EXERCISE 161. Record the following payment transactions for Letzelter Co. of Saint John,NB, during the month of November. Set up a Cash Payments Journal(page CP6) with these headings: Date,...
-
The CECL model:Multiple ChoiceConsiders historical experience but not forecasts of the future.Recognizes bad debts when it is probable that an economic sacrifice has occurred.Allows a company to use...

Study smarter with the SolutionInn App


