Question: Identify which compounds below possess a molecular dipole moment and indicate the direction of that dipole moment: a. b. c. d. :i: i: .CI: II
Identify which compounds below possess a molecular dipole moment and indicate the direction of that dipole moment:
a.
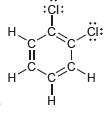
b.
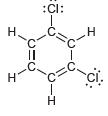
c.
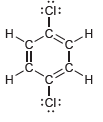
d.
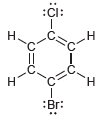
:i: i: .CI: II .C. .C. H. :: :Cl: . .C. .C. di: . CHO
Step by Step Solution
★★★★★
3.34 Rating (166 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a b c There is no mo... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


