In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure
Question:
Figure 15.27
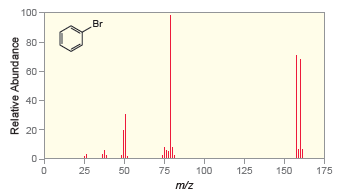
a) Does this fragment contain Br? Explain your reasoning.
b) Draw the cationic fragment that represents the base peak.
Transcribed Image Text:
100 Br 80 60 40- 20 50 25 75 100 125 150 175 m/z Relative Abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
a This fragment is ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
The mass spectrum of 1-butanol shows an intense daughter ion peak at m/z 5 31. Explain how this peak might arise.
-
Identify the expected base peak in the mass spectrum of 2, 2, 3-trimethylbutane. Draw the fragment associated with this peak, and explain why the base peak results from this fragment.
-
As speed increases for an object in free fall, does acceleration increase also?
-
What is the role of specialists and traders on the New York Stock Exchange?
-
A surgeon places an electrode on a portion of your brain and stimulates it. Immediately, your right wrist involuntarily twitches. The doctor has most likely stimulated a portion of the __________area...
-
Nancy Carpenter Optical Dispensary purchased supplies on account for \($1,200\). Two weeks later, the business paid half on account. Requirements 1. Journalize the two transactions for Nancy...
-
1. What is the macro and industry environment in the Southeast Asian region for the entrance of new budget airlines? What opportunities and challenges are associated with this environment? 2. How...
-
H Show that 60 000 H W = dn (2) From WV where W, transmitted load, kN H = power, kW (15 Points) d = gear diameter, mm n = speed, rev/min
-
Palins Muffler Shop has one standard muffler that fits a large variety of cars. The shop wishes to establish a periodic review system to manage inventory of this standard muffler. Use the information...
-
Show how you would use a Grignard reaction to prepare each compound below. a. b. c. d. e. f. OH
-
Suppose that the pen-making industry is perfectly competitive. Also suppose that each current firm and any potential firms that might enter the industry all have identical cost curves, with minimum...
-
Use @RISK to draw a uniform distribution from 400 to 750. Then answer the following questions. a. What are the mean and standard deviation of this distribution? b. What are the 5th and 95th...
-
Assume a city of \(1,000,000\) people, \(60 \%\) of whom are willing to pay \(\$ 1\) maximum (each) to clean up pollution. The rest of the population is wealthier and is willing to pay \(\$ 100\)...
-
Consider the case of a rival bad. Would efficiency require that a Pigovian fee be levied on the producer of the bad and the receipts given to the consumers as compensation? Does it matter if the bad...
-
In the Amoco Cadiz oil spill off of Northern France, fisheries, water fowl, and beaches were temporarily negatively impacted. List five types of consumers, identify the nature of goods these...
-
Zhejiang Corporation sells customized stage lighting equipment for use in the entrainment industry. Zhejiang has a broad dealer network. One dealer, Min Chen, obtained a large contract with a ship...
-
You are the mayor of the small town of Wasilla and a landowner has offered to sell you 1,000 hectares of woodland for \(\$ 2,000,000\). You are very tempted because of the wildlife which live there...
-
Voltaire Enterprises statement of financial position at December 31, 2019, is presented below. During 2020, the following transactions occurred. 1. On January 1, 2020, Voltaire issued 1,500 20 par,...
-
Listed below are common types of current liabilities, contingencies, and commitments: a. Accounts payable b. Bank loans and commercial paper c. Notes payable d. Dividends payable e. Sales and excise...
-
Draw a Newman projection along the C2-C3 bond of the following conformation of 2, 3-dimethylbutane, and calculate total strainenergy:
-
Identify the functional groups in the following substances, and convert each drawing into a molecular formula (red = O, blue =N): (a) (b) Phenylalanine Lidocaine
-
Give IUPAC names for the following alkanes, and convert each drawing into a skeletal structure: (a) (b) (c) (d)
-
The Cited paper is about Sustainability in Organizations: A case of Public companies in Nigeria. What are the prevailing themes that link the academic articles? (What is the common topic matter among...
-
1. This week covers IT Project Management. History has shown many projects fail to deliver within budget or do not meet customer expectations. The text does not address this item in any great deal...
-
According to the Josephine Institute, rank the core ethical values in order. After ranking them, discuss how you would apply them when faced with a situation where you must choose between right and...

Study smarter with the SolutionInn App


