N-Acetylazoles undergo hydrolysis more readily than regular amides. Suggest a reason for the enhanced reactivity of N-acetylazoles
Question:
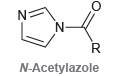
Transcribed Image Text:
N: 'N- N-Acetylazole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The lone pair of the nitrogen atom in this case is participa...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Pyrrole undergoes electrophilic aromatic substitution more readily than benzene, and mild reagents and conditions are sufficient. These reactions normally occur at the 2-osition rather than the...
-
A surface for which the electrostatic potential is negative delineates regions in a molecule that are subject to electrophilic attack. It can help you to rationalize the widely different chemistry of...
-
The explosion of an atomic bomb releases many radioactive isotopes, including strontium-90. Considering the location of strontium in the periodic table, suggest a reason for the fact that this...
-
What is unrealistic about the domino fault model?
-
What competitive advantage or disadvantage might an eBooks only textbook publisher have relative to McGraw-Hill, Pearson, and other large international publishers?
-
Solve the given systems of equations by using the inverse of the coefficient matrix. Use a calculator to perform the necessary matrix operations and display the results and the check. See Example 4....
-
Adverse and sudden developments in political and legal systems can create country risk. Suggest three examples of such adverse developments. LO.1
-
At December 31, 2017, the available-for-sale debt portfolio for Steffi Graf, Inc. is as follows. On January 20, 2018, Steffi Graf, Inc. sold security A for $15,100. The sale proceeds are net of...
-
chap 7 hw 1 Check my work 1 1 Exercise 7-2 (Algo) First Stage Allocation (L07-2) 10 points SecuriCorp operates a fleet of armored cars that make scheduled pickups and deliveries in the Los Angeles...
-
From soap to soup, Unilever markets a wide range of personal care products, foods, and household cleaners under popular brands like Dove, Bertolli, Lipton, Lux, Axe, Sunsilk, Surf, and Omo. Two...
-
Propose a mechanism for the following transformation, and explain how you could use an isotopic labeling experiment to verify your proposed mechanism: [H,SO,] R.
-
Dimethylformamide (DMF) is a common solvent: (a) The 1 H NMR spectrum of DMF exhibits three signals. Upon treatment with excess LAH followed by water, DMF is converted into a new compound that...
-
A subsidy is a negative tax through which the government gives people money instead of taking it from them. If the government applied a $1.05 specific subsidy instead of a specific tax in Figure...
-
Section Three Answer the questions below 1.While pulling out of her driveway, Bethany becomes distracted by a bee and strikes Melanie, who is riding past on a bicycle. Bethany suffers serious injury...
-
A __________ is a schedule periodic check of a specific process behavior. Question 1Answer A. Widget B. Dashboard C. Monitor D. Process ID
-
1. Was VAAF contractually obligated to pay Chad for refraining from smoking? 2. Was there consideration to support its promise to pay $500? 3. Are there other facts you need to know to make that...
-
Presented here are the comparative balance sheets of Hames Incorporated at December 31, 2023 and 2022. Sales for the year ended December 31, 2023, totaled $1,700,000.%0D%0A%0D%0AHAMES...
-
McDonald's conducts operations worldwide and is managed in two primary geographic segments: US, and International Operated Markets, which is comprised of Australia, Canada, France, Germany, Italy,...
-
In Problems 2532, graph the line that contains the point P and has slope m. P= (0, 3); slope undefined
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
Give structures for each of the following compounds. C8H10O: IR, 3150-3600 cm-1 (strong, broad); NMR, 1.17 (3/7, t, 7 = 8 Hz); 2.58 (2/7, q, / = 8 Hz); 6.0 (1/7, broad singlet, disappears with D20...
-
Give structures for each of the following compounds. C8H10O: IR, 3150-3600 cm-1 (strong, broad); NMR, 1.17 (3/7, t, 7 = 8 Hz); 2.58 (2/7, q, / = 8 Hz); 6.0 (1/7, broad singlet, disappears with D20...
-
How would you distinguish mesitylene (1, 3, 5-trimethylbenzene) from isopropylbenzene (cumene) by 13C NMR spectroscopy?
-
3. The nominal interest rate compounded monthly when your $7,000 becomes $11,700 in eight years is ________
-
An investor can design a risky portfolio based on two stocks, A and B. Stock A has an expected return of 21% and a standard deviation of return of 39%. Stock B has an expected return of 14% and a...
-
Advanced Small Business Certifica Drag and Drop the highlighted items into the correct boxes depending on whether they increase or decrease Alex's stock basis. Note your answers- you'll need them for...

Study smarter with the SolutionInn App


