Predict the products of each of the following reactions: a. b. c. d. e. f. g. h.
Question:
a.
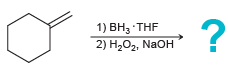
b.
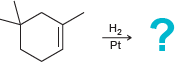
c.
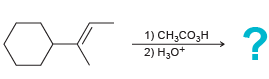
d.
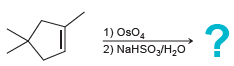
e.
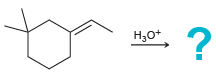
f.
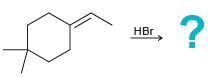
g.
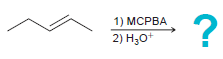
h.
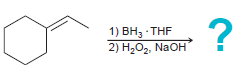
i.
Transcribed Image Text:
1) BH3 THF 2) H2O2, NaOH На Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a b c d ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. i. j. HCI CH2CH3 1. CH3CH2MgBr 1. CH3CH2MgBr excesS CH,CH,COCH 2. H20 ot.cum 1. LiAIH4 NO 2. H20 catalytic Ht + CH...
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. CH CH3 CH CH H2S04 CH,CH CHCH CH ,504 CH CH-CCH CH3 OH ' CH3O CH3OH 1. TsCl/pyridine 2. NaCEN CH3 OH (CH2CH2CH2hCuli CI
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. hv CH3 H3C CH3 A DHHD H3C
-
Using algebra, find the slope and y-intercept of the line represented by each of the following equations. (a) 7x + 3y = 6 (b) 10y = 5x (c) (2y 3x)/2 = 4 (d) 1.8x + 0.3y 3 = 0 (e) 1/3x = -2 (f) 11x...
-
The following activities were observed for a mold operator: a) operator removes molded piece from die, b) walks to bench, puts the piece into box and the puts the box on a pallet, c) returns to...
-
What is the electrochemical potential difference when the intracellular [Ca2+] = 1 M and the extracellular [Ca2+] = 1 mM? Assume = -100 mV (inside negative) and T = 25C.
-
What is the major difference between a standard accounting system and a voucher system?
-
Call Options Hedging Foreign Currency Debt Taking advantage of lower interest rates in the United Kingdom, Carlton Inc., a U.S. firm, borrowed 2,000,000 on July 1, 2012, to be repaid in one year....
-
Which type of deduction does not require the employer to have the employee's written permissi O Company-compulsory deductions O Legal deductions O Voluntary deductions O All of the above
-
Consider the below table of 6 observations. Values of X1 and X2 are used to predict the outcome Y. When Y is 1 the outcome is considered to be positive. What is the Gini impurity of the given data...
-
Why is a correlation c oefficient never greater than 1 (or less than 1)?
-
The rate at which two methyl radicals couple to form ethane is significantly faster than the rate at which two tert-butyl radicals couple. Offer two explanations for this observation.
-
Do marginal costs always consist only of variable costs?
-
Jane McDonald is a 28-year-old solicitor at a prestigious law firm in Sydney. Jane rents a studio apartment near the Sydney central business district, paying $650 per week. She estimates her other...
-
helens investment of 500 has grown at a rate of 13% compounded annually , for 45 years. what is the maturity value of her investment
-
Division D operates as on investment centre. The book value of the non-current assets is $ 83,000 but their replacement value is estimated to be $ 98,000. Working capital in the division has a value...
-
Discuss the molecular mechanisms underlying cellular apoptosis and its significance in maintaining tissue homeostasis and eliminating aberrant cells in multicellular organisms.
-
Evaluate Nike Co. in terms of the ratio analysis, horizontal analysis and vertical analysis. In words address the following: What do the ratios calculated over the two-year period tell us about the...
-
John Williams (birthdate August 2, 1976) is a single taxpayer. Johns earnings and withholdings as the manager of a local casino for 2019 are reported on his Form W-2: Johns other income includes...
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
Draw compounds that meet these requirements: (a) A primary alcohol (b) A tertiary alcohol (c) A secondary alkyl chloride (d) A secondary amine
-
What is wrong with the name given for these compounds? Provide the correct name for each. a) c) 18 2-Ethyl-2-pentene CHCHCH 3-Cyclohexylpropane 1-Chloro-2-cyclopentene OH 2-2-Methylbutylcyclopentanol...
-
Name these compounds: a) ) g) 1) k) - OH NH b) CI d) f) CH=CHCHC=CH h) Br j) O OCH 3
-
Garden Depot is a retailer that is preparing its budget for the upcoming fiscal year. Management has prepared the following summary of its budgeted cash flows: Total cash receipts Total cash...
-
(b) After analyzing the accounts, journalize the adjusting entries that were made on July 31. (List all debit entries before credit entries.Credit account titles are automatically indented when the...
-
Use the following information: a. Beginning cash balance on March 1, $81,000. b. Cash receipts from sales, $306,000. c. Budgeted cash payments for direct materials, $136,000. d. Budgeted cash...

Study smarter with the SolutionInn App


