Predict which of the following two compounds will undergo an E2 reaction more rapidly: CI
Question:
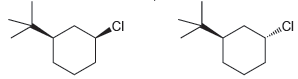
Transcribed Image Text:
CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 65% (20 reviews)
Because of the bulky tertbutyl group the first compou...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would a structural effect that destabilizes the acid component of a conjugate acid-base pair affect its acidity? Use your analysis to predict which of the following two compounds is more basic....
-
Which of the following two compounds would eliminate HBr more rapidly in basic solutions? CH2Br or CH2Br
-
(a) Two stereoisomers of a bromodecalin are shown. Although the difference between these stereoisomers may seem trivial, one isomer undergoes elimination with KOH much faster than the other. Predict...
-
Elaborate the basic functioning of a hydraulic lift. Include the terms pressure, density, and buoyant force in the explanation. Give one example of a hydraulic lift you have encountered.
-
A natural deposit of soil is found to have a water content of 20% and to be 90% saturated. What is the void ratio of this soil?
-
In Exercises determine whether the statement is true or false. If it is false, explain why or give an example that shows it is false. If () = g() for = 0, /2, and 3/2, then the graphs of r = () and...
-
2. Locate the Web site for a municipality (city, county, town, parish, etc.) in which your college or university is located. Is the municipalitys financial report available on the Web site? If so,...
-
A production manager at a Contour Manufacturing plant has inspected the number of defective plastic molds in 5 random samples of 20 observations each. Following are the number of defective molds...
-
Please answer the following accounting question. Oriole Googal owns a garage and is contemplating purchasing a tire retreading machine for $18,820. After estimating costs and revenues, Oriole...
-
Sage Inc. bought 40% of Adams Corporations outstanding common stock on January 2, 20X1, for $400,000. The carrying amount of Adamss net assets at the purchase date totaled $900,000. Fair values and...
-
The Organization for African Unity has vigorously opposed the export of hazardous waste from industrialized countries to developing nations. They call this practice toxic terrorism. Explain.
-
Compare integrated pest management to integrated waste management. How does each reduce potential damage to the environment?
-
Choice Foods Inc. uses activity-based costing to determine product costs. For each activity listed in the left column, match an appropriate activity base from the right column. You may use items in...
-
In the following circuit, the supply voltage is 12V. Suppose VIN = +7 V, the output voltage will be, VIN VOUT R1 10 R2 10
-
Find the derivative of the function f(x)=(7x-8)". Find the derivative of the function y=- 2x x+6 Find the derivative of the function y = In Find the derivative of the function f(t): Find the...
-
Use the information below to answer questions 29 - 39. Make a table similar to examples 12 and 13 to help answer the questions. Ten persons of students at ACM are nursing students. Forty percent of...
-
Abby Industries, Inc. has the following capital structure. Type Amount Rate of Return Mortgages (debt) $25,000,000 7% Bonds (debt) 180,000,000 9% Common stock (equity) 100,000,000 10% Preferred stock...
-
Review the network of stakeholders Choose five different stakeholders and provide examples of why a project manager would need to negotiate with that stakeholder. FIGURE 10.1 Network of Stakeholders...
-
Simplify each expression. Assume that all variables represent nonzero real numbers. (3pq)q 6pq4
-
The ultimate goal of Google, Bing, and other consumer search engines is to provide users with search listings that contain useful information on the topic of their search. What recommendations would...
-
Predict the products from reaction of 1-hexyne with the following reagents: (a) 1 equiv HBr (b) 1 equiv Cl2 (c) H2, Lindlar catalyst (d) NaNH2 in NH3, then CH3Br (e) H2O, H2SO4, HgSO4 (f) 2 equiv HC1
-
Predict the products from reaction of 5-decyne with the following reagents: (a) H2 Lindlar catalyst (b) Li in NH3 (c) 1 equiv Br2 (d) BH3 in THF, then H2O2, OH (c) H2O, H2SO4, HgSO4 (f) Excess H2,...
-
Predict the products from reaction of 2-hexyne with the following reagents: (a) 2 equiv Br2 (b) 1 equiv HBr (c) Excess HBr (d) Li in NH3 (e) H2O, H2SO4, HgSO4
-
Provide a graph chart or data with sample numbers indicating Valuing Stocks and Bonds?
-
I just need help with part b. It says that the answer is not complete and some are wrong. So can you kindly fix it for me and give me the full answers as it says the answer is "not complete". Thank...
-
What is Coke's average ownership percentage in its equity method investments? Goodwill is 7000 Calculate the firm's current ratio (current assets/current liabilities). Calculate the current ratio...

Study smarter with the SolutionInn App


