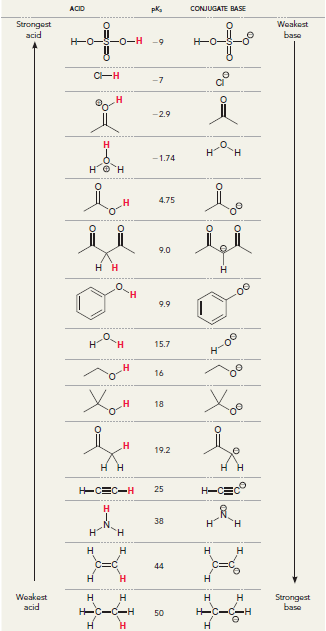
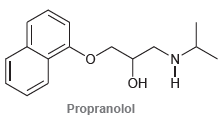
Propanolol is an antihypertensive agent (used to treat high blood pressure). Using the following table, identify the
Question:
Transcribed Image Text:
ACID pka CONJUGATE BASE Weakest Strongest acid for baso -0- -0- -7 -2.9 н "н -1.74 4.75 9.0 нн 9.9 н 15.7 Ay-00 16 18 н 19.2 H-CE 25 -CEC-H 38 н н н н Н н 44 н Н Н Woakest acid н н Н н Strongest base H-C-C -с-С-Н 50 Н н O EC I-C N. Он Н Propranolol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
pK16 ...View the full answer

Answered By

Shivani Dubey
Hello guys, I am very good with the Advance mathematics, Calculus, Number theory, Algebra, Linear algebra, statistics and almost all topics of mathematics. I used to give individual tutoring students in offline mode but now want to help students worldwide so I am joining here. I can also give solutions in various coding languages of mathematics and software like mathematica, R, Latex, Matlab, Statistica, etc.
feel free to ask any doubt regarding mathematics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Many naturally occurring nitrogen compounds and many nitrogen-containing drugs are better known by common names than by their systematic names. A few of these follow. Write a structural formula for...
-
Below is the structure of rilpivirine, a promising new anti-HIV drug that combats resistant strains of HIV. Its ability to side-step resistance will be discussed in the upcoming chapter. (a) Identify...
-
Valproic acid, used to treat seizures and bipolar disorder, is composed of C, H, and O. A 0.165-g sample is combusted in an apparatus such as that shown in Figure 3.14. The gain in mass of the H2O...
-
In Exercises explain why Rolle's Theorem does not apply to the function even though there exist a and b such that (a) = (b). f(x) H [1,1]
-
Now you are going to do some calculations for the delivery times for each week using the mean delivery time which is 23 minutes and the standard deviation which was 20 minutes. (a) Describe the...
-
Without consulting the text, write the Hamiltonian operator for the lithium atom without using any sum symbols. Assume an infinitely heavy nucleus (and omit spin-orbit interaction).
-
Production-volume variances arise with normal-absorption and standard-absorption costing, but not with actual costing. Explain.
-
RATIO ANALYSIS OF COMPARATIVE FINANCIAL STATEMENTS Refer to the financial statements in Problem 24-8B. REQUIRED Calculate the following ratios and amounts for 20-1 and 20-2 (round all calculations to...
-
If a pencil weighs 0.085 kilograms, it weighs g.
-
Determine the volume (in millilitres) of 0.2 M sodium iodate, 0.2 M sodium bisulfite and deionised water required to prepare the two solutions needed to perform an iodine clock reaction that changes...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (b) C (d) H. -S -3- (f) X H' `H.
-
L-dopa is used in the treatment of Parkinsons disease. Using the follwoing table, identify the four most acidic protons in the compound, and then arrange them in order of increasing acidity (two of...
-
A copper wire has a 25-m resistance at 20oC. When the wire is carrying a current, heat produced by the current causes the temperature of the wire to increase by 20oC. (a) What is the change in the...
-
3 01:27:46 Book Required: Katy Williams is the manager of Blue Light Arcade. The company provides entertainment for parties and special events. For each of the following transactions, give the...
-
3. The population (in thousands) in Meridian, Idaho over the last twenty years is listed in the table below. (Data from world populationreview.com) Use the t-values given in the table for your...
-
Provide C++ Program to copy strings .
-
Provide C++ Program to find the frequency of characters in a string .
-
Write C++ Program to find largest element of array .
-
Show that LRU is m-competitive for any sequence of n page requests, where m is the size of the memory cache.
-
What services are provided by the provincial and territorial governments?
-
When propylbenzene reacts with chlorine in the presence of UV radiation, the major product is 1-chloro-1-phenylpropane. Both 2-chloro-1-phenylpropane and 3-chloro- 1-phenylpropane are minor products....
-
Show how the following compounds could be synthesized from phenylacetylene (C6H5CCH): (a) 1-phenylpropyne, (b) 1-phenyl-1-butyne, (c) (Z )-1-phenylpropene, and (d) (E )-1-phenylpropene. Begin each...
-
Can you suggest an explanation that accounts for the fact that the radical polymerization of styrene (C6H5CHwCH2) to produce polystyrene occurs in a head-to-tail fashion, rather than the head-to-head...
-
Way Cool produces two different models of air conditioners. The company produces the mechanical systems in its components department. The mechanical systems are combined with the housing assembly in...
-
Hatch Manufacturing produces multiple machine parts. The theoretical cycle time for one of its products is 30 minutes per unit. The budgeted conversion costs for the manufacturing cell dedicated to...
-
The objective of the Additional Refundable Tax on Investment Income is to discourage the use of a Canadian controlled private corporation to defer taxes on investment income. . True False

Study smarter with the SolutionInn App


