Starting with acetylene, show the reagents you would use to prepare the following compounds: (a) 1-Butyne (b)
Question:
(a) 1-Butyne
(b) 2-Butyne
(c) 3-Hexyne
(d) 2-Hexyne
(e) 1-Hexyne
(f) 2-Heptyne
(g) 3-Heptyne
(h) 2-Octyne
(i) 2-Pentyne
(j)
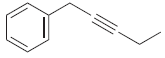
(k)
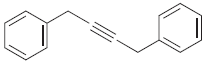
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
a b c d ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of the azo dye called alizarine yellow R (below). Show the reagents you would use to prepare this compound via an azo coupling process. .N. N' O2N
-
Using ammonia as your source of nitrogen, show the reagents you would use to prepare each of the following amines: (a) (b) (c) (d) (e) (f) Z
-
Identify the reagents you would use to prepare the following compound via a Robinson annulation. . 0=
-
Consider the market for milk. Draw a supply curve and a demand curve (is the demand curve elastic on inelastic). Label the equilibrium price and quantity. Suppose that the production of milk causes...
-
XYZ Company uses activity-based pricing. The company prices their products by charging customers the direct cost by using a markup of 35% on the direct cost plus adding service cost that are...
-
Find the vector with the given magnitude and the same direction as u. Magnitude ||v|| = 6 Direction u = (0, 3)
-
What are the four assumptions on which economic-order quantities are based? For what kind of items are these assumptions valid? When are they not? LO.1
-
Tip Top Company sells umbrellas suited for small and large picnic tables. Based on the advice of its accountant, Tip Top is considering whether to adopt an activity-based costing system. To evaluate...
-
The new Designer Shoes was expected to sell for $ 104 per unit and had projected sales of 4700 units in the first year, with a projected (Most-Likely scenario) 15.0 % growth rate per year for...
-
Huffman codes compress text by assigning the characters that occur at the highest frequency the shortest possible codes. In this encoding scheme, no code can be a prefix of another. For example, if...
-
An alkyne with molecular formula C 4 H 6 was treated with ozone followed by water to produce a carboxylic acid and carbon dioxide. Draw the expected product when the alkyne is treated with aqueous...
-
Preparation of 2,2-dimethyl-3-octyne cannot be achieved via alkylation of acetylene. Explain.
-
On January 1, 2021, Poplar Fabricators Corporation agreed to grant its employees two weeks of vacation each year, with the stipulation that vacations earned each year can be taken the following year....
-
Brice Looney owns a small retail ice cream parlor. He is considering expanding the business and has identified two attractive alternatives. One involves purchasing a machine that would enable Mr....
-
A positively charged particle initially at rest on the ground moves \(4.0 \mathrm{~m}\) upward in \(2.00 \mathrm{~s}\). If the particle has a chargeto-mass ratio of \(10 \mu \mathrm{C} / \mathrm{g}\)...
-
Central States Telecom provides communication services in Iowa, Nebraska, the Dakotas, and Montana. Central States purchased goodwill as part of the acquisition of Sheldon Wireless Company, which had...
-
Shown below is selected information from the financial records of Merris Corporation as of December 31: Required a. Determine which of the above items will appear on the statement of cash flows and...
-
Pippa runs a photographic studio specializing in black and white portrait photography. Clients book a one hour studio session and are entitled to receive two large photographs of their choice from...
-
Use a graphing utility to sketch the graph of f(x) = x 3 + 3x 2 5x + 11. Then sketch the graph of g(x) = (x + 1) 3 + 3(x + 1) 2 5(x + 1) + 11 on the same axes. What function h(x) would have a graph...
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
Thiamin, or vitamin Bi, contains a positively charged five-membered nitrogen?sulfur heterocycle called a thiazolium ring. Explain why the thiazolium ring is aromatic NH2 . Thiamin , H Thiazolium ring
-
Show the relative energy levels of the seven molecular orbitals of the Cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the...
-
Azulene, a beautiful blue hydrocarbon, is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance form of azulene in addition to thatShown. Azulene
-
Show that the convexity for a zero coupon bond with m payments per year is (m) n(n + -)(1+ m m
-
Abdul Canarte , a Central Bank economist, noticed that the total group purchasing basket of goods (CPI) has gone from $149,740.00 to $344,460.00 in 8 years. With monthly compounding, what is the...
-
ABC Corporation expects sales next year to be $50,000,000. Inventory and accounts receivable (combined) will increase $8,000,000 to accommodate this sales level. The company has a profit margin of 6...

Study smarter with the SolutionInn App


