Using 1-propanol as your only source of carbon, propose an efficient synthesis for each of the following
Question:
(a)
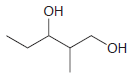
(b)
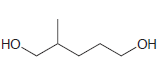
(c)
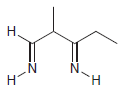
Transcribed Image Text:
ОН ОН но но.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a b c OH ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using 2-propanol as your only source of carbon, show how you would prepare 2-methyl-2-pentanol.
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 1-bromobutane.
-
What is the present value of $9,000 received: a. Twenty eight years from today when the interest rate is 10% per year? b. Fourteen years from today when the interest rate is 10% per year? c. Seven...
-
The risk-free rate, KRF, is 6 percent and the market risk premium, (KM - KRF), is 5 percent. Assume that required returns are based on the CAPM. Your $1 million portfolio consists of $700,000...
-
Why is an American option always worth more than its intrinsic value? (As an example, recall that the intrinsic value at time t for the call option is max(S(t) K; 0).)
-
Suppose you are considering acquiring a house by assuming the remainder of the existing 30-year mortgage on the property. If there are 327 fixed monthly payments of $1,041.23 left to be paid, with...
-
Kansas Corp., an American company, has a payment of 5 million due to Tuscany Corp. one year from today. At the prevailing spot rate of 0.90 /$, this would cost Kansas $5,555,556, but Kansas faces the...
-
Blue Company was formed on July 1, 2018. It was authorized to issue 298,000 shares of $10 par value common stock and 102,100 shares of 8% $25 par value, cumulative and nonparticipating preferred...
-
A small British computer-game firm, Eidos Interactive PLC, stunned the U.S.- and Japan-dominated market for computer games when it introduced Lara Croft, an Indiana Jones-like adventuress. The...
-
Refer to the illustrative example of Chapter 7 where we fitted the Cobb Douglas production function to the manufacturing sector of all 50 states and the District of Columbia for 2005. The results of...
-
Refer to Example 7.4. For this problem the correlation matrix is as follows: a. Since the zero-order correlations are very high, there must be serious multicollinearity. Comment. b. Would you drop...
-
How does the issue of double taxation arise when a corporation decides between making a distribution to a shareholder employee as a dividend or compensation?
-
What is the best 3D printer for consumer use? Why?
-
a. What are the purposes of the auditor's final assessment of materiality and audit risk? b. Distinguish between the terms known misstatement, likely misstatement, and aggregate likely misstatement.
-
What is the best 3D printer for industrial use? Why?
-
Identify several types of cash accounts that should be included as cash balances on the balance sheet and several others that should not.
-
Will 3D printing be bigger than the internet? Why?
-
The rational-actor paradigm assumes that people do NOT a. Act rationally. b. Ase rules of thumb. c. Act optimally. d. Act self-interestedly.
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
Identify each of the following compounds from their spectra. (a) Compound A: molecular mass 113; gives a positive hydroxamate test; IR 2237, 1733, 1200 cm-1; proton NMR: 1.33 (3H, t, J = 7 Hz), ...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
q1: q2: help and explain The Museum of History is preparing for its annual appreciation dinner for contributing members. Last year, 650 members attended the dinner. Tickets for the dinner were $26...
-
This is part 2. The following information is from the Oregon Revised Statutes, Chapter 673, for Tax Consultants and Tax Preparers. Click the following link to access the reading material for part 2...
-
The manager explained that this practice created a pool of money that could be refunded to departments when they were having financial difficulties. It's also known that department managers receive a...

Study smarter with the SolutionInn App


