Consider the equilibrium CO(g) + H2O(g) ???? CO2(g) + H2(g). At 1150. K, the composition of the
Question:
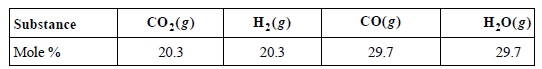
a. Calculate KP and ΔGoR at 1150. K.
b. Given the answer to part (a), use the ΔHof of the reaction species to calculate ΔGoR at 298.15 K. Assume that ΔHR° is independent of temperature.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





