In this problem, you will use the variational method to find the optimal 1s wave function for
Question:
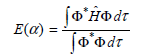
with respect to α.
a. Show that
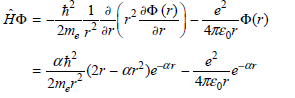
b. Obtain the result
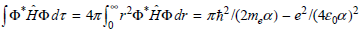
Using the standard integrals in the Math Supplement (Appendix A)
c. Show that
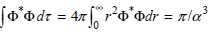
Using the standard integrals in the Math Supplement.
d. You now have the result E (α) = h2α2 / (2me) €“ e2α / (4πε0). Minimize this function with respect to α and obtain the optimal value of α
e. Is E(αoptimal) equal to or greater than the true energy? Why?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





