The Grotrian diagram in Figure 22.7 shows a number of allowed electronic transitions for He. Which of
Question:
a. 1s2 1S †’ 1s2p 1P
b. 1s2p 1P †’ 1s3s 1S
c. 1s2s 3S †’ 1s2p 3P
d. 1s2p 3P †’ 1s3d 3D
Figure 22.7
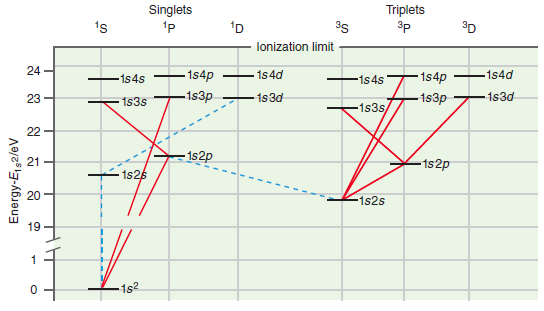
Transcribed Image Text:
Singlets Triplets 1s 35 3D lonization limit 24 - 1s4p 1s4d -1s4d - 1s4p -1s4s 1s4s -1s3p 1s3d 1s3d -1s3p 23 1s3s 1s3s, 22 - 152p 21 -1s2p 1s26 -1s2s 19 1s2 Energy-E92/eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
a 1s 2 1 S 1s2p 1 P No splitting because each term consists of a singl...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The figure below represents part of the emission spectrum for a one- electron ion in the gas phase. All the lines result from electronic transitions from excited states to the n = 3 state. a. What...
-
The following is an energy- level diagram illustrating three different electronic transitions in the Bohr hydrogen atom. a. Explain why the energy levels get closer together as they increase. Provide...
-
The energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one anti-bonding. In ethylene there is a pair of...
-
Reading material Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Product: The Online Offer. E-Marketing , 9, 206 - 228. Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Price: The Online Value....
-
Determine the angle between the diagonal of a cube and the diagonal of its base, as shown in the figure. a a a
-
In Problems 106111, solve each equation. Express irrational solutions in exact form. Inx = (lnx)
-
True or False. The correlation coefficient is a measure of the LO9 strength of the linear relationship between x and y.
-
This year Burchard Company sold 40,000 units of its only product for $25 per unit. Manufacturing and selling the product required $200,000 of fixed manufacturing costs and $325,000 of fixed selling...
-
80 3. a) A Japanese soft-drink company is planning to establish a subsidiary company in India to produce mineral water. Based on the estimated annual sales of 40,000 bottles of mineral water cost...
-
The following data represent the number of appointments made per 15-minute interval by telephone solicitation for a lawn-care company. Assume these are population data. Number of Frequency...
-
In the Na absorption spectrum, the following transitions are observed: 4p 2 P 3s 2 S = 330.26 nm 3p 2 P 3s 2 S = 589.593 nm, 588.996 nm 5s 2 S 3p 2 P = 616.073 nm, 615.421 nm Calculate the...
-
List the quantum numbers L and S that are consistent with the following terms: a. 4 S b. 4 G c. 3 P d. 2 D
-
In the expres sion var result = sourceString.localeCompare(compareString), what are the possible return values and what does each mean?
-
solve for x 4 . 0 a 2 = 2 . 0 a x
-
BUSINESS SOLUTIONS Comparative Balance Sheets March 3 1 , 2 0 2 2 December 3 1 , 2 0 2 1 Assets Cash $ 8 4 , 7 8 7 $ 5 7 , 8 7 2 Accounts receivable 2 4 , 2 6 7 5 , 0 6 8 Inventory 6 1 4 0 Computer...
-
Solve:z-18=-103.
-
Complete the social penetration exercise and post your reactions in the discussion. PIRATION Purpose: 1. To help you understand the breadth and depth of self-disclosure. 2. To help you see the...
-
The implicit equation of x = sin ( t ) and y = 2 cos ( t ) is:
-
In Exercises 4152, give the center and radius of the circle described by the equation and graph each equation. Use the graph to identify the relations domain and range. (x + 2) + (y + 2) = 4
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Distinguish between semi-empirical, ab initio, and density functional theory methods of electronic structure determination.
-
What information does the term symbol 1 D 2 provide about the angular momentum of an atom?
-
Show by explicit integration that (a) Hydrogenic 1s and 2s orbitals, (b) 2p x and 2p y orbitals are mutually orthogonal.
-
Slow Roll Drum Co. is evaluating the extension of credit to a new group of customers. Although these customers will provide $198,000 in additional credit sales, 13 percent are likely to be...
-
Wendell's Donut Shoppe is investigating the purchase of a new $39,600 conut-making machine. The new machine would permit the company to reduce the amount of part-time help needed, at a cost savings...
-
1.Discuss the challenges faced with Valuing Stocks and Bonds. As part of this discussion, how will the selected item be implemented in an organization and its significance? 2. Discuss how Valuing...

Study smarter with the SolutionInn App


