According to Figure 2, which of the following compounds would most typically be found only in rocks
Question:
According to Figure 2, which of the following compounds would most typically be found only in rocks at the moderate stage?
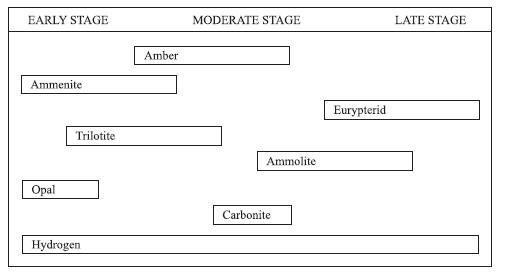
F. Ammoenite
G. Eurypterid
H. Carbonite
J. Clay
Transcribed Image Text:
EARLY STAGE Ammenite Opal Trilotite Hydrogen Amber MODERATE STAGE Ammolite Carbonite Eurypterid LATE STAGE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The best answer is H According to Figure 2 the only compound that i...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
1. In the classical model, it is thought that the long-run: A. and short-run aggregate supply curves are both upward sloping. B. aggregate supply curve is vertical and the short-run aggregate supply...
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Jillian operates a sporting goods store in a rented location at a shopping mall. She is insured under a CGL policy with the following limits: Indicate the dollar amount, if any, that Jillians insurer...
-
Plattsburg Plastics Corporation manufactures a variety of plastic products, including a series of molded chairs. The three models of molded chairs, which are all variations of the same design, are...
-
A rigid tank contains nitrogen gas at 227C and 100 kPa gage. The gas is heated until the gage pressure reads 250 kPa. If the atmospheric pressure is 100 kPa, determine the final temperature of the...
-
The spines on a head of lettuce mirror the Fibonacci sequence. If a head of lettuce contains 13 spines, approximately how many spines would be found on the next inside layer?
-
Suppose the own price elasticity of demand for good X is 2, its income elasticity is 3, its advertising elasticity is 4, and the cross-price elasticity of demand between it and good Y is 6....
-
please solve it perfectly and also please read the instructions that what kind of answer they are asking only only write final answer un the form that they are asking Determine the velocity vector...
-
Cast fossils are formed when material is placed into a depression in a rock near the surface. They are formed at very high temperatures and low pressures. According to Figure 1, cast fossils are most...
-
Which of the following graphs best represents the relationship between time elapsed and saturation of the rocks observed in Experiment 1? Experiment 1 In a laboratory, a scientist wanted to determine...
-
(a) Find the series radius and interval of convergence. For what values of x does the series converge (b) Absolutely, (c) Conditionally? 8 n=1 (x - 1)n n 3n
-
On December 31, 2011, Chang Company had the following normal account balances in its general ledger. Use this information to prepare a trial balance. Common Stock $25,000 Salaries Expense 16,000...
-
Are there any cases you can find where a supplemental pleading was allowed because new facts have been alleged? What were the new facts?
-
Research a past California case in which you believe a removal would have changed the outcome of the case. Using your knowledge of the potential advantages of transferring a case already filed in the...
-
Reston Manufacturing Corporation produces a cosmetic product in three consecutive processes. The costs of Department | for May 2016 were as follows: Department | handled the following units during...
-
Implement the nave Bayes classifier in the programming language of your choice, and use it to classify pages of text, based on which words appear on the page. To do this, you will first need to train...
-
Fred Burton, the owner of a small tennis club in Wichita, Kansas, feels that a demand exists for indoor courts that presently is not being served. He is considering employing a marketing research...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
If the demand for perfume is inelastic above and below the present price, should the price be raised? Why or why not?
-
If the demand for shrimp is highly elastic below the present price, should the price be lowered?
-
Discuss what factors lead to inelastic demand and supply curves. Are they likely to be found together in the same situation?
-
PLEASE OPEN IMAGE AND ANSWER ALL REQUIREMENTS Requirement 1 . Calculate the price per hour that New York Temps should charge based on Mason's proposal. = Price per hour Requirement 2 . On the basis...
-
dig in to the three methods we can use to value inventory: FIFO, LIFO and average cost. There are pros and cons to each method. What is key is that we are using a consistent method in valuing...
-
Fixed factory overhead cost $ 3 9 , 0 0 0 Fixed selling and administrative costs 7 , 4 0 0 Variable direct materials cost per unit Variable direct labor cost per unit 5 . 0 1 Variable factory...

Study smarter with the SolutionInn App


