Answered step by step
Verified Expert Solution
Question
1 Approved Answer
1. part A & B The evaporation of a 130nm film of n-pentane If the initial surface coverage is 8.71016 molecules /cm2, how long will
1. part A & B 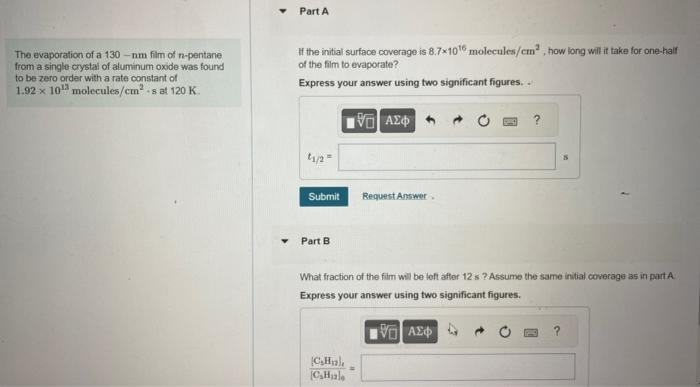
The evaporation of a 130nm film of n-pentane If the initial surface coverage is 8.71016 molecules /cm2, how long will it take for one-half from a single crystai of aluminum oxide was found of the film to evaporate? to be zero order with a rate constant of 1.921013molecules/cm2-sat120K. Part B What fraction of the film will be left after 12 s ? Assurne the same initial coverage as in part A. Express your answer using two significant figures 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


