Answered step by step
Verified Expert Solution
Question
1 Approved Answer
1. Which among the following can serve as buffer system for a solution with pH = 3.0? 2. Which among the following has the highest
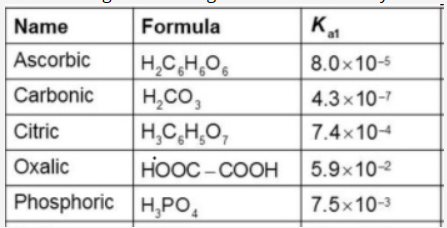
1. Which among the following can serve as buffer system for a solution with pH = 3.0?
2. Which among the following has the highest acidity?
3. Which among the following has the highest pH?
\begin{tabular}{|l|l|l|} \hline Name & Formula & Kat \\ \hline Ascorbic & H2C6H6O6 & 8.0105 \\ \hline Carbonic & H2CO3 & 4.3107 \\ \hline Citric & H3C6H5O7 & 7.4104 \\ \hline Oxalic & HOOCCOOH & 5.9102 \\ \hline Phosphoric & H3PO4 & 7.5103 \\ \hline \end{tabular}Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


