Answered step by step
Verified Expert Solution
Question
1 Approved Answer
1. You have an electron in a two-dimensional quantum box that is 8.43 nm wide and 8.43 nm. Calculate the energies of the lowest 3
1. You have an electron in a two-dimensional quantum box that is 8.43 nm wide and 8.43 nm. Calculate the energies of the lowest 3 states, and write down their wavefunctions.
2. The Bohr radius is given by ?0 h2 / [? e2 me]. Look up the constants and calculate the value of this expression.
- Starting from the units of the constants you used, show that your Bohr radius has units of meters.
3. Show that the g(?) we guessed in Lecture #32 is indeed a solution to the ? part of the Schroedinger equation for the hydrogen atom.
4. Calculate the mean value of r for an electron in the ground state of a hydrogen atom. The mean value is given by
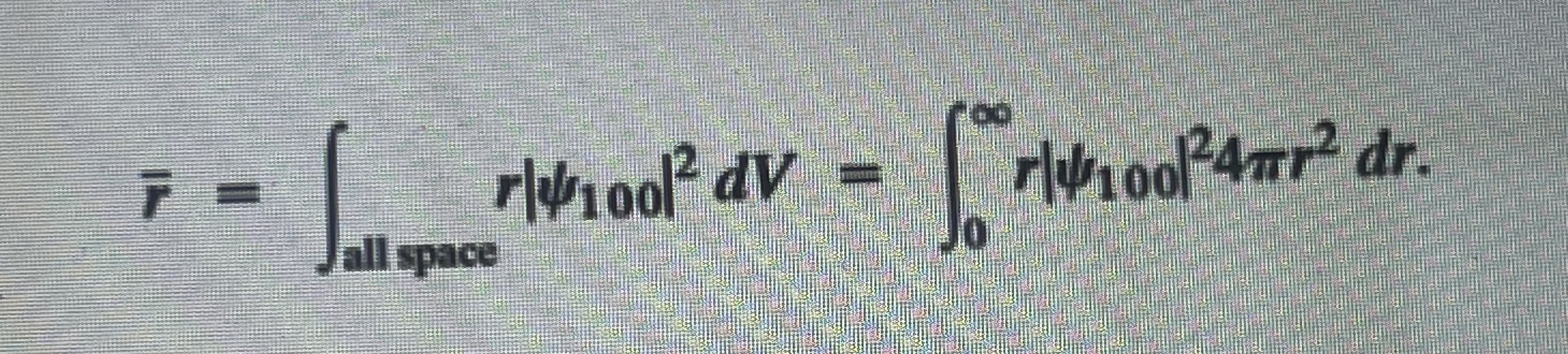
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


