Answered step by step
Verified Expert Solution
Question
1 Approved Answer
11. An incompressible liquid is kept in a container having a weightless piston with a hole. A capillary tube of inner radius 0.1 mm
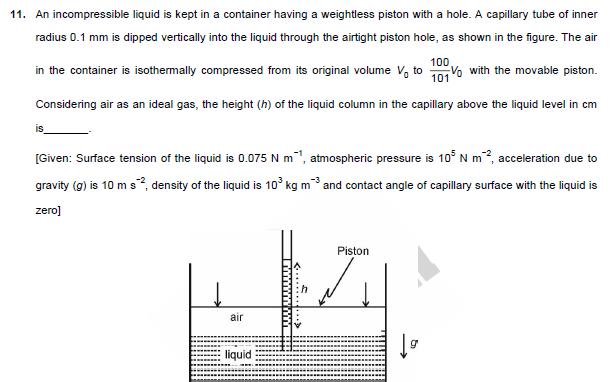
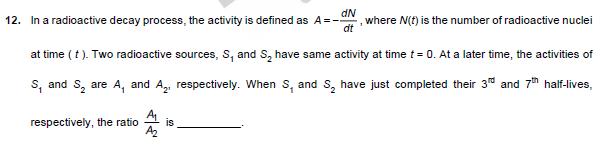
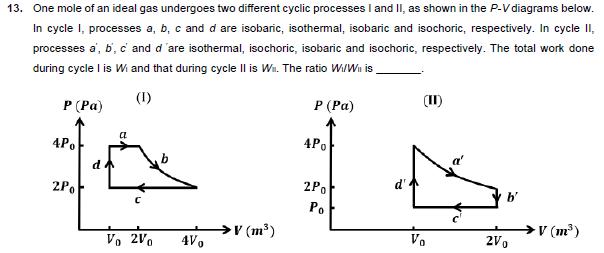
11. An incompressible liquid is kept in a container having a weightless piston with a hole. A capillary tube of inner radius 0.1 mm is dipped vertically into the liquid through the airtight piston hole, as shown in the figure. The air 100 in the container is isothermally compressed from its original volume Voto 1010 with the movable piston. Considering air as an ideal gas, the height (h) of the liquid column in the capillary above the liquid level in cm [Given: Surface tension of the liquid is 0.075 N m, atmospheric pressure is 105 N m, acceleration due to gravity (g) is 10 m s, density of the liquid is 103 kg m and contact angle of capillary surface with the liquid is zero] air liquid Piston 12. In a radioactive decay process, the activity is defined as A=- dN dt ,where N(t) is the number of radioactive nuclei at time (t). Two radioactive sources, S, and S have same activity at time t = 0. At a later time, the activities of S, and S are A, and A, respectively. When S, and S have just completed their 3rd and 7th half-lives, respectively, the ratio A2 is 13. One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the P-V diagrams below. In cycle I, processes a, b, c and d are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes a, b, c and d 'are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is Wi and that during cycle II is W. The ratio W/W is (II) (I) P (Pa) a 4Po d 2Po P (Pa) 4Po 2Po Po V (m) Vo 2V0 Vo 2Vo 4V0 V (m)
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


