Question
19.2. A chemistry lab contains an eye wash station as shown in the figure. Water flows steadily through the eye wash station, entering at
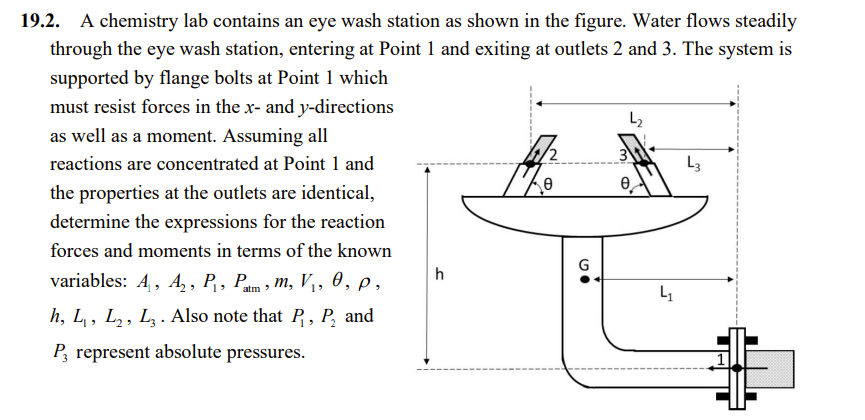
19.2. A chemistry lab contains an eye wash station as shown in the figure. Water flows steadily through the eye wash station, entering at Point 1 and exiting at outlets 2 and 3. The system is supported by flange bolts at Point 1 which must resist forces in the x- and y-directions as well as a moment. Assuming all reactions are concentrated at Point 1 and the properties at the outlets are identical, determine the expressions for the reaction forces and moments in terms of the known variables: A, A, P, Pam, m, V, 0, p, h, L, L, L3. Also note that P, P and P, represent absolute pressures. 2 h 4/2 G L e L L3
Step by Step Solution
3.40 Rating (144 Votes )
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Fundamentals Of Momentum Heat And Mass Transfer
Authors: James Welty, Gregory L. Rorrer, David G. Foster
6th Edition
1118947460, 978-1118947463
Students also viewed these Mechanical Engineering questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



