Answered step by step
Verified Expert Solution
Question
1 Approved Answer
2. Casein molecules in milk have some glutamic acid residues that change their charge based on the pH of their surroundings. This is one factor
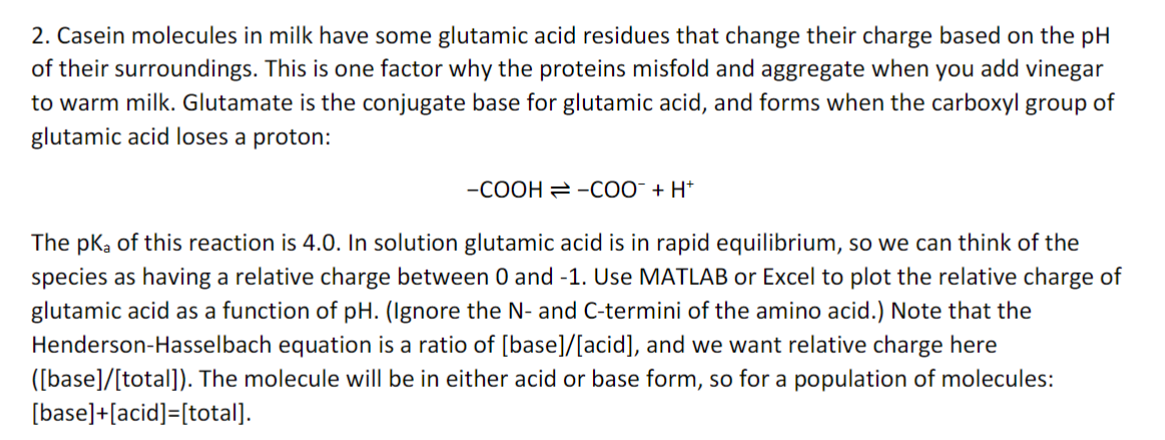
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


