(2 points) Carbon dating uses Carbon-14, a radioactive isotope of carbon, to measure the age of...
Fantastic news! We've Found the answer you've been seeking!
Question:
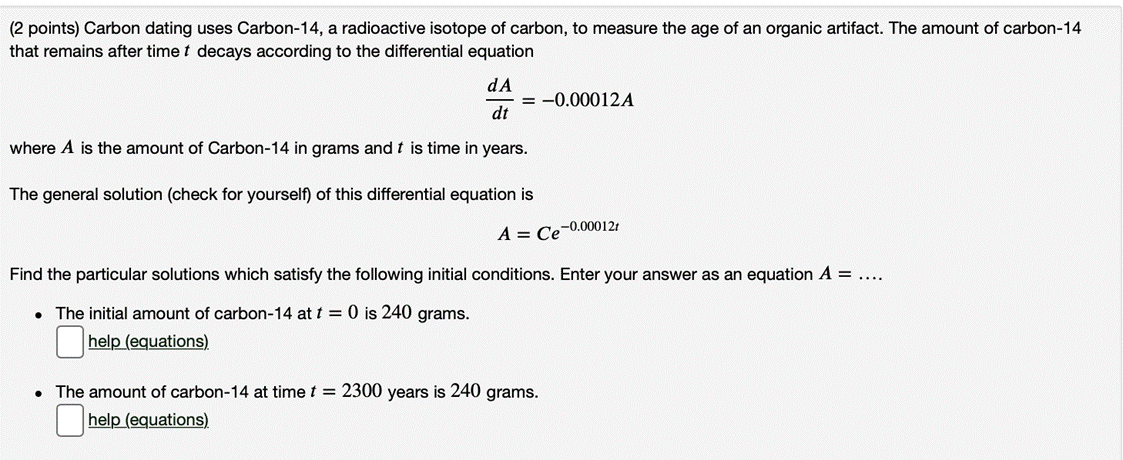


Transcribed Image Text:
(2 points) Carbon dating uses Carbon-14, a radioactive isotope of carbon, to measure the age of an organic artifact. The amount of carbon-14 that remains after time t decays according to the differential equation dA = -0.00012A dt where A is the amount of Carbon-14 in grams and t is time in years. The general solution (check for yourself) of this differential equation is A = Ce-0.00012t Find the particular solutions which satisfy the following initial conditions. Enter your answer as an equation A = .... • The initial amount of carbon-14 at t = 0 is 240 grams. help (equations) • The amount of carbon-14 at time t = 2300 years is 240 grams. |help (equations) You are contracted to install Netscape Navigator software on all the PCs of company X After doing half the work, you found that company X is not paying Netscape for the copies you are installing You notified company X's contact that they are out of compliance with Netscape licensing requirement, but got no response • What do you do? Computer Ethics - Case Studies Example 1: You are asked to write a program to print tags for a sale. Your boss asks you to put tags that have a price 10% higher, with a 10% discount marking it back to original price. Do you do this? Example 2: I You wrote a software that matches the requirement your company was given. But, requirements are so bad that you know the software will not match the actual needs • Should you say anything? • Should your company say anything? Even if it would mean loss of future contracts? Part 2 CSC 42 Computing Ethics 18 (2 points) Carbon dating uses Carbon-14, a radioactive isotope of carbon, to measure the age of an organic artifact. The amount of carbon-14 that remains after time t decays according to the differential equation dA = -0.00012A dt where A is the amount of Carbon-14 in grams and t is time in years. The general solution (check for yourself) of this differential equation is A = Ce-0.00012t Find the particular solutions which satisfy the following initial conditions. Enter your answer as an equation A = .... • The initial amount of carbon-14 at t = 0 is 240 grams. help (equations) • The amount of carbon-14 at time t = 2300 years is 240 grams. |help (equations) You are contracted to install Netscape Navigator software on all the PCs of company X After doing half the work, you found that company X is not paying Netscape for the copies you are installing You notified company X's contact that they are out of compliance with Netscape licensing requirement, but got no response • What do you do? Computer Ethics - Case Studies Example 1: You are asked to write a program to print tags for a sale. Your boss asks you to put tags that have a price 10% higher, with a 10% discount marking it back to original price. Do you do this? Example 2: I You wrote a software that matches the requirement your company was given. But, requirements are so bad that you know the software will not match the actual needs • Should you say anything? • Should your company say anything? Even if it would mean loss of future contracts? Part 2 CSC 42 Computing Ethics 18
Expert Answer:
Answer rating: 100% (QA)
To find the particular solutions that satisfy the given initial conditions we can substitute the values into the general solution of the differential equation and solve for the constant C 1 Initial co... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A radioactive isotope of copper decays as follows: Starting with 84.0 g of 64Cu, calculate the quantity of 64Zn produced after 18.4 h. 6Cu 64, 64Zn + -iB = 12.8 h
-
Can 14/6C dating be used to measure the age of stone walls and tablets of ancient civilizations? Explain
-
A radioactive isotope of mercury, 197Hg, decays to gold, 197Au, with a disintegration constant of 0.0108 6-t. (a) Calculate the half-life of the 167Hg. What fraction of a sample will remain at the...
-
Why do we use the absolute value of x or of g(x) in the derivative formulas for the natural logarithm?
-
Consider the problem above, but now each bidder has a value of either $60 or $80. What reserve price should the auctioneer set, and what is the expected revenue from auctioning the item with and...
-
The Kramer Corporation's marketing manager calculates a regression, where the quantity demanded of the firm's product (designated as "C1") is the dependent variable and the price of the product...
-
Dennis Williams is projecting the coming years net income potential for Williams Paint. The paint is sold for \($15.00\) a gallon. Variable costs per gallon are \($10.00\), and annual fixed costs are...
-
Multiple Choice 1. The present value of the minimum lease payments should be used by the lessee in the determination of a (an) Capital Operating Lease Liability Lease Liability a. Yes ......No b. Yes...
-
Explain how your chosen five documents relate to laws and regulations on construction in Aotearoa New Zealand. Include discussion points that are relevant to the laws or regulations in relation to...
-
(A) Use the appropriate location procedure to determine the optimal coordinates of a new machine based on the following assumptions. (Please show the detaila of your work: Not just the final answer):...
-
Suppose that you are the new CEO at Memorial Hospital. Me- morial is a nonprofit hospital with 300 beds and is located in a busy metropolitan area directly adjacent to a large university. Memorial is...
-
Select a valid company in Canada and make an Marketing presentation on it with references. Every point should be with detailed information ?
-
A monopoly with constant marginal costs mc = $20 has two potential groups of customers, whose demands are Q1=100-0.8p and Q2=100 p respectively and who cannot trade with each other. i.(5%) If the...
-
Why would you as a business owner be interested in liquidity ratios. Explain in details.
-
adjusted trial balance of 022: Qotba Company Adjusted Trial Balance As at 31st December 2022 CCOUNT TITLE Cash Accounts Receivable Office Supplies DEBIT QAR 164,800 CREDIT 45,000 Prepaid Insurance...
-
As you know, public goods are goods or services that everyone can enjoy. Even nonpayers can enjoy the benefits of public goods. Do you believe the power grid should be considered a public good?...
-
2. (40 pts) Describe the "supply chain plan for Apple. Note that the plan should be based on the instructions provided for the term project. Page Break
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
Aspirin or acetyl salicylic acid is synthesized by reacting salicylic acid with acetic anhydride: (a) How much salicylic acid is required to produce 0.400 g of aspirin (about the content in a...
-
Determine the pH of
-
For the reaction (a) Predict the enthalpy of reaction from the average bond enthalpies in Table 9.4. (b) Calculate the enthalpy of reaction from the standard enthalpies of formation (see Appendix 3)...
-
In the preparation of an analytic flowchart for a payroll processing application, which of the following symbols could be used to indicate the payroll data? (a) connector symbol (b) decision symbol...
-
In the preparation of an analytic flowchart, which of the following symbols should be used when flowlines are broken due to a page limitation? (a) terminal symbol (b) connector symbol (c) manual...
-
Assume trays are plug flow, and repeat Problem 16.D21 parts a and \(b\). In addition, calculate \(\mathrm{E}_{\mathrm{pt}}\) for the three mole fractions \(\mathrm{x}_{\mathrm{W}}=0.48,0.36\), and...

Study smarter with the SolutionInn App


