Answered step by step
Verified Expert Solution
Question
1 Approved Answer
20. The diagram below represents a large-scale fractional distillation plant used to separate the components A, B, C and D in a mixture mixture
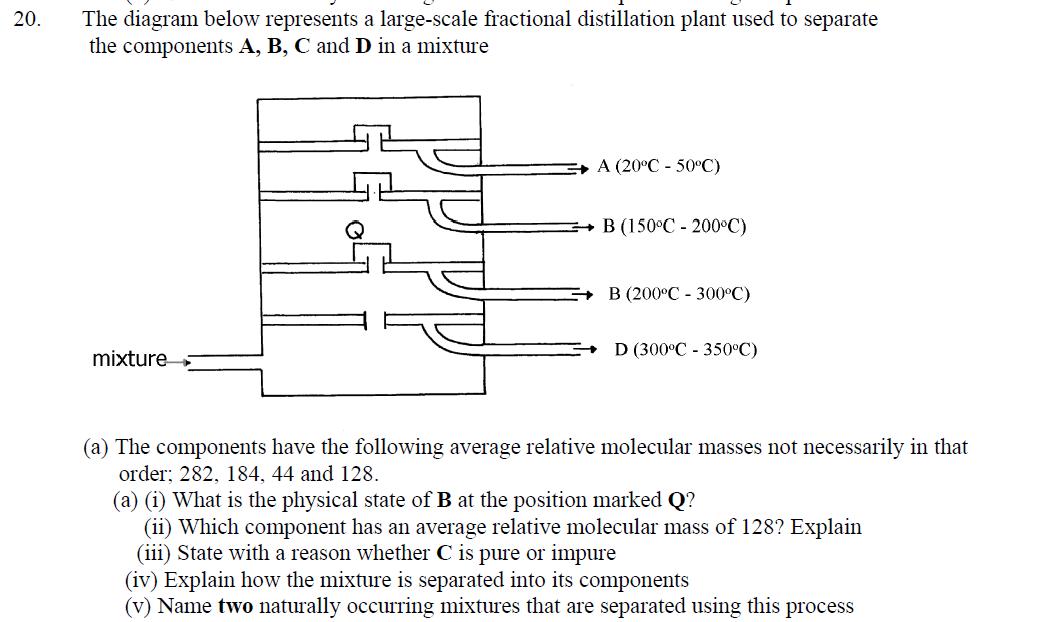

20. The diagram below represents a large-scale fractional distillation plant used to separate the components A, B, C and D in a mixture mixture A (20C -50C) B (150C 200C) B (200C - 300C) D (300C 350C) (a) The components have the following average relative molecular masses not necessarily in that order; 282, 184, 44 and 128. (a) (i) What is the physical state of B at the position marked Q? (ii) Which component has an average relative molecular mass of 128? Explain (iii) State with a reason whether C is pure or impure (iv) Explain how the mixture is separated into its components (v) Name two naturally occurring mixtures that are separated using this process 221 21. a) The table below gives information about the major constituents of crude oil. Study it and answer the questions that follow: Constituent Gases Boiling point C Below 40 Petrol Kerosene Diesel Lubricating oil 40-175 175-250 250-350 350-400 Bitumen Above 400 i) Which of the constituents of crude has molecules with the highest number of carbon atoms? Explain ii) Name the process you would use to separate a mixture of petrol and diesel and explain how the separation takes place iii) Explain why the constituents of crude oil do not have a sharp boiling point iv) Name the gas that is likely to be a constituent of crude oil and write its formula b) i) What condition could cause a poisonous gas to be formed when kerosene is burnt. Explain ii) Give one use of bitumen
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


