Answered step by step
Verified Expert Solution
Question
1 Approved Answer
2.18 At 25C we have for rhombic sulfur: Gf=0,S=31.880.17J/Kmol; and for monoclinic sulfur: Gf=63J/mol,S=32.550.25J/Kmol. Assuming that the entropies do not vary with temperature, sketch the
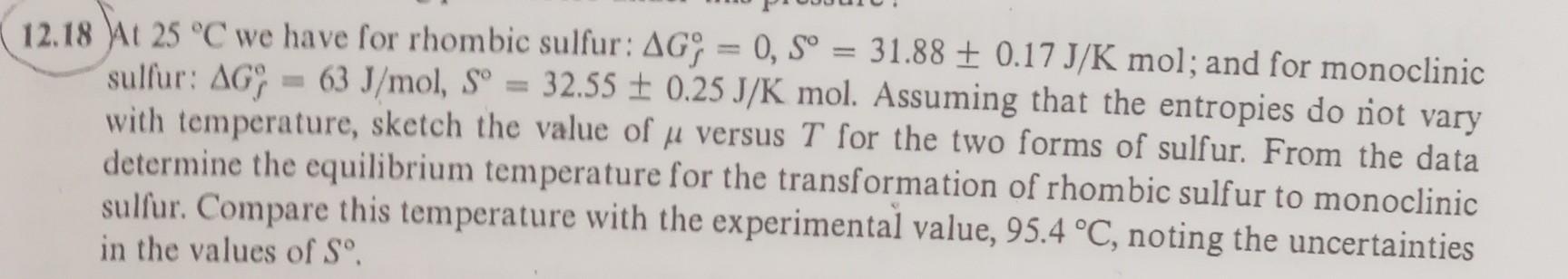
2.18 At 25C we have for rhombic sulfur: Gf=0,S=31.880.17J/Kmol; and for monoclinic sulfur: Gf=63J/mol,S=32.550.25J/Kmol. Assuming that the entropies do not vary with temperature, sketch the value of versus T for the two forms of sulfur. From the data determine the equilibrium temperature for the transformation of rhombic sulf ur to monoclinic sulfur. Compare this temperature with the experimental value, 95.4C, noting the uncertainties in the values of S
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


