Answered step by step
Verified Expert Solution
Question
1 Approved Answer
25. parts a and b The equiliorium constant, K, for the following reaction E.4.56102 at 639K. COCl2(g)=Co(g)+Cl2(g) An equilibrium maxture of the three gases in
25. parts a and b 
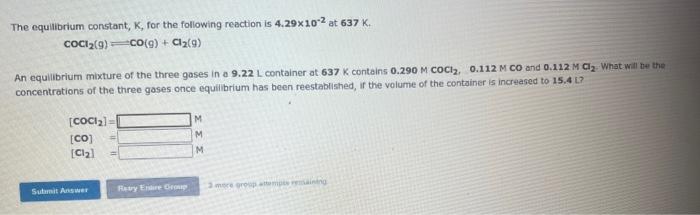
The equiliorium constant, K, for the following reaction E.4.56102 at 639K. COCl2(g)=Co(g)+Cl2(g) An equilibrium maxture of the three gases in a 16.3L container at 639K contains 0.264MCOCl2.0.110MCO and 0.110M Cl. . What wit be the concentrations of the three gases once equilibrium has been reestablished, if the equilibrium micture is compressed at constant temperature to a volume of 9.62L? [COCl2]={CO]=[Cl2]= The equilibrium constant, K, for the following reaction is 4.29102 at 637K. COCl2(g)=CO(g)+Cl2(g) An equilibrium mixture of the three gases in a 9.22L container at 637K contains 0.290MCOCl,0.112MCO and 0.122MCl2. What will bet the concentrations of the three gases once equilibrium has been reestablished, if the volume of the container is increased to 15.4 Li? [COCl2]=[[[CO]=[Cl2]= 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


