Answered step by step
Verified Expert Solution
Question
1 Approved Answer
2nd Edition Introductory Physics V3 revised.pdf 243 2nd Edition Introductory Physics V3 revised.pdf 243 244 245 246 247 24: Lab 19' 345 Electric Charge and
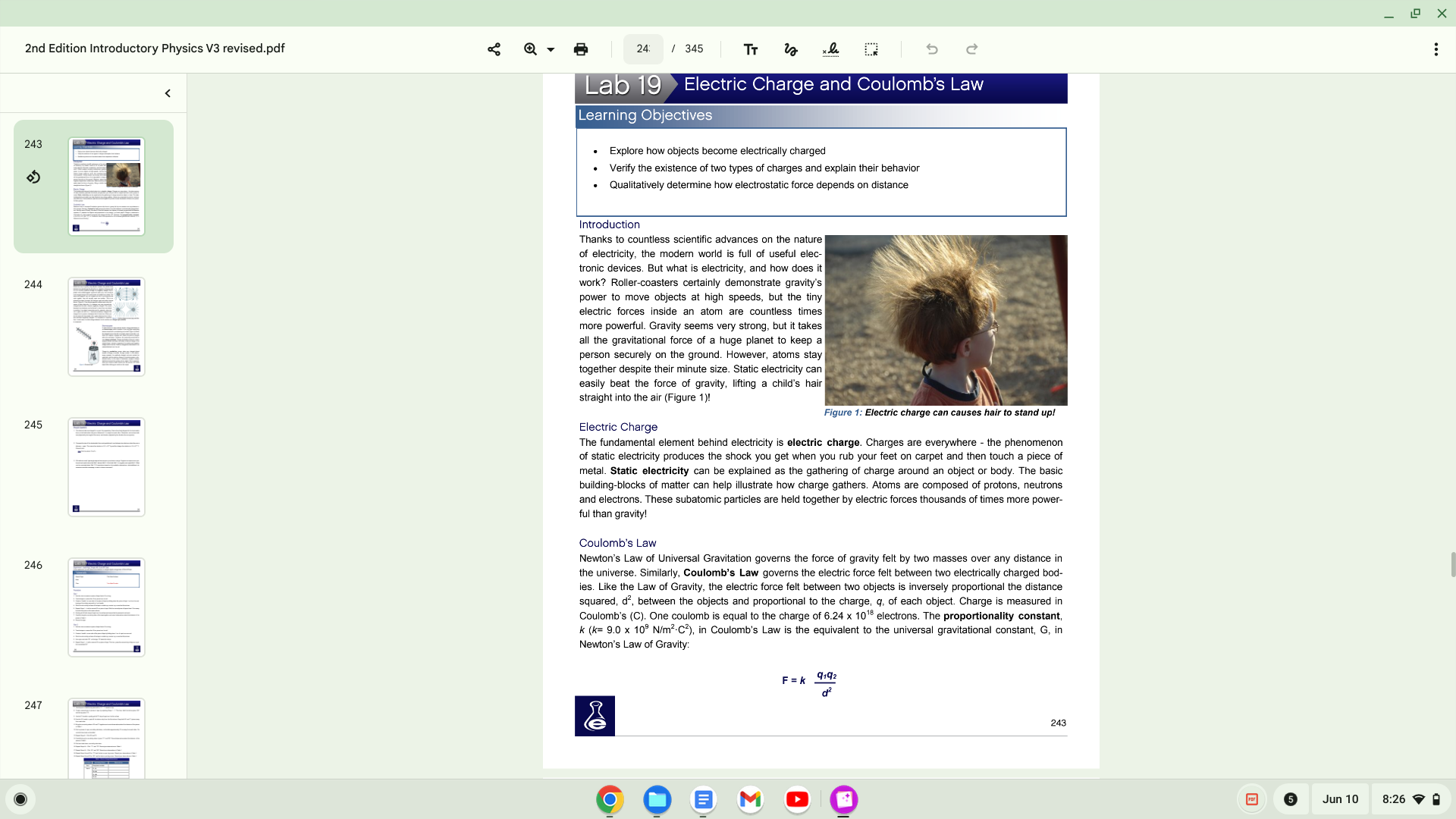

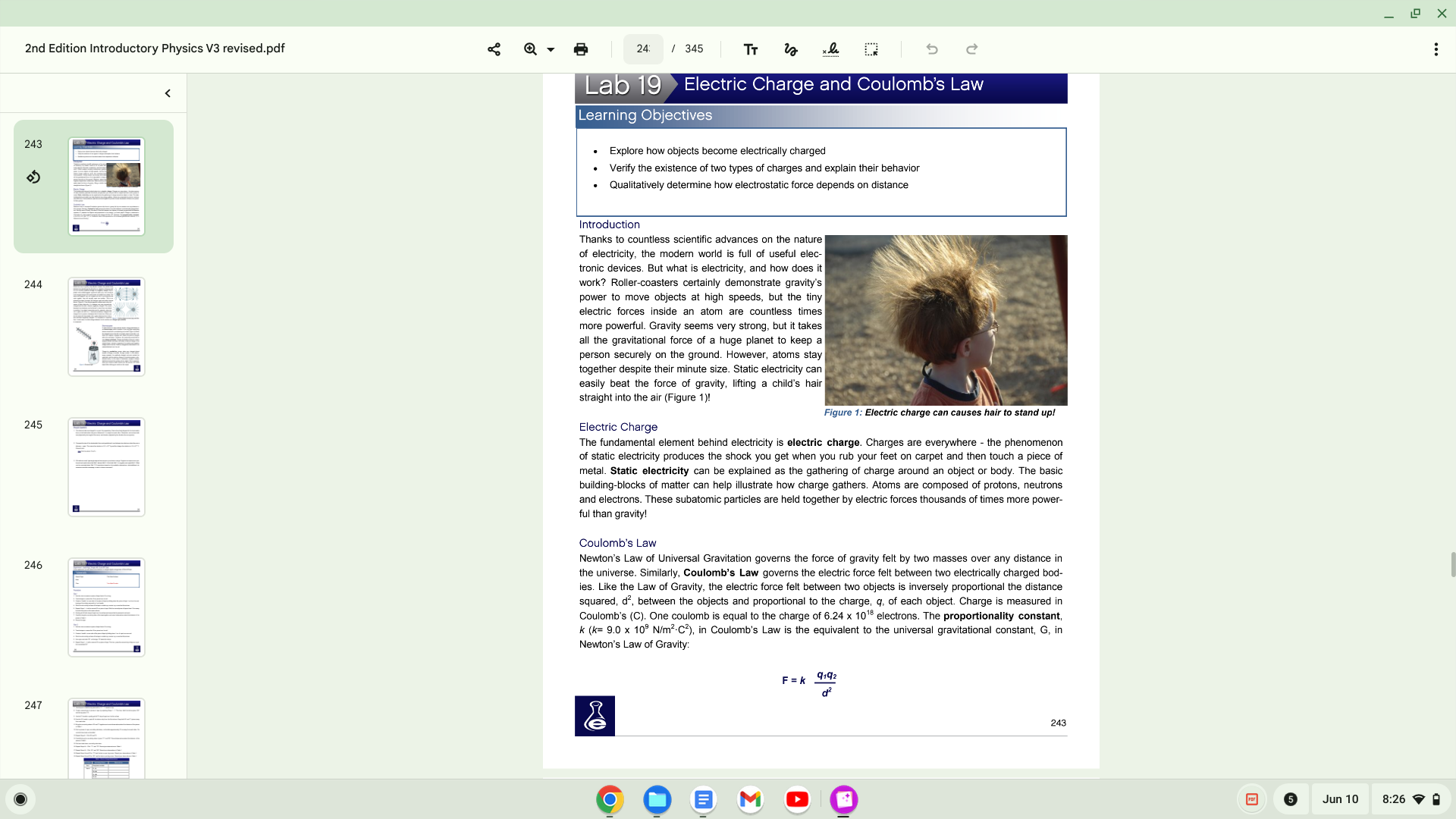

2nd Edition Introductory Physics V3 revised.pdf 243 244 245 246 247 24: Lab 19' 345 Electric Charge and Coulomb's Law Learning Objectiv Explore how objects become electrically charged Verify the existence of tv.'0 types of charges and explain their behavior Qualitatively determine how electrostatic force depends on distance Introduction Thanks to countless scientific advances on the nature of electricity, the modem world is full of useful elec- tronic devices But what is electricity, and how does it work? Roller-coasters certainly demonstrate gravity's power to move objects at high speeds, but the tiny electric forces inside an atom are countless times more powerful. Gravity seems very Strong, but it takes all the gravitational force of a huge planet to keep a person securely on the ground. However, atoms stay despite their minute size. Static electricity can easily beat the force of gravity, lifting a child's hair straight into the air (Figure 1)! Electric Charge Figure I: Electric charge can causes hair to stand upl The fundamental element behind electricity is electric charge. Charges are everywhere - the phenomenon of static electricity produces the shock you get when you rub your feet on carpet and then touch a piece of metal. Static electricity can be explained as the gathering of charge around an object or body. The basic building-blocks of matter can help illustrate how charge gathers. Atoms are composed of protons, neutrons and electrons. These subatomic particles are held together by electric forces thousands of times more power- ful than gravity! Coulomb's Law Newton's Law of Universal Gravitation governs the force of gravity felt by two masses over any distance in the universe. Similarly, Coulomb's Law governs the electric force felt between two electrically charged bod- 'es Like the Law of Gravity, the electric force felt between objects is inversely proportional the distance squared, d2, between the objects and proportional to the charge, q, of each object. Charge is measured in Coulomb's (C). One coulomb is equal to the charge of 6.24 x 1018 electrons. The proportionality constant, k 9.0 x 109 N/m2C2), in Coulomb's Law is the equivalent to the universal gravitational constant, G, in Newton's Law of Gravity. q,q2 243 O O Jun 10 x 8:26 a 2nd Edition Introductory Physics V3 revised.pdf 243 244 245 246 247 24: Lab 19' 345 Electric Charge and Coulomb's Law Learning Objectiv Explore how objects become electrically charged Verify the existence of tv.'0 types of charges and explain their behavior Qualitatively determine how electrostatic force depends on distance Introduction Thanks to countless scientific advances on the nature of electricity, the modem world is full of useful elec- tronic devices But what is electricity, and how does it work? Roller-coasters certainly demonstrate gravity's power to move objects at high speeds, but the tiny electric forces inside an atom are countless times more powerful. Gravity seems very Strong, but it takes all the gravitational force of a huge planet to keep a person securely on the ground. However, atoms stay despite their minute size. Static electricity can easily beat the force of gravity, lifting a child's hair straight into the air (Figure 1)! Electric Charge Figure I: Electric charge can causes hair to stand upl The fundamental element behind electricity is electric charge. Charges are everywhere - the phenomenon of static electricity produces the shock you get when you rub your feet on carpet and then touch a piece of metal. Static electricity can be explained as the gathering of charge around an object or body. The basic building-blocks of matter can help illustrate how charge gathers. Atoms are composed of protons, neutrons and electrons. These subatomic particles are held together by electric forces thousands of times more power- ful than gravity! Coulomb's Law Newton's Law of Universal Gravitation governs the force of gravity felt by two masses over any distance in the universe. Similarly, Coulomb's Law governs the electric force felt between two electrically charged bod- 'es Like the Law of Gravity, the electric force felt between objects is inversely proportional the distance squared, d2, between the objects and proportional to the charge, q, of each object. Charge is measured in Coulomb's (C). One coulomb is equal to the charge of 6.24 x 1018 electrons. The proportionality constant, k 9.0 x 109 N/m2C2), in Coulomb's Law is the equivalent to the universal gravitational constant, G, in Newton's Law of Gravity. q,q2 243 O O Jun 10 x 8:26 a
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


