Question
3- In an experiment, we drop a metal of mass 100g and temperature of 100C a colorimeter has a water of mass 200 g
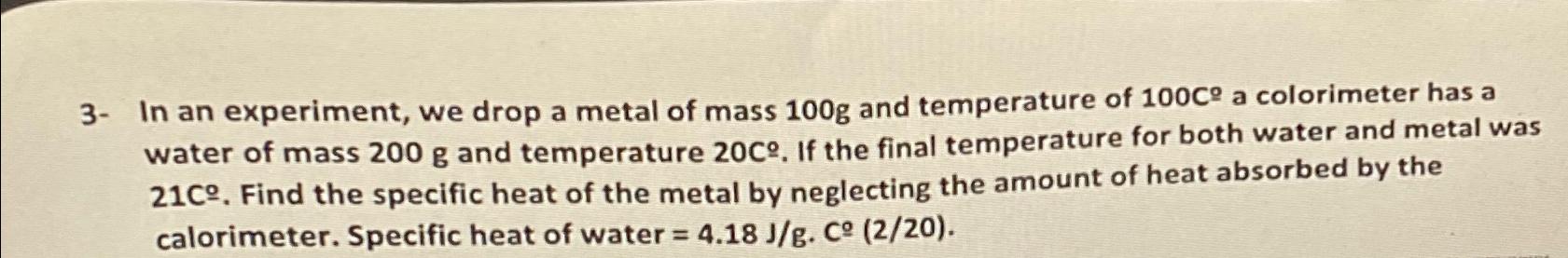
3- In an experiment, we drop a metal of mass 100g and temperature of 100C a colorimeter has a water of mass 200 g and temperature 20C. If the final temperature for both water and metal was 21C. Find the specific heat of the metal by neglecting the amount of heat absorbed by the calorimeter. Specific heat of water = 4.18 J/g. C (2/20).
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
College Physics
Authors: Jerry D. Wilson, Anthony J. Buffa, Bo Lou
7th edition
9780321571113, 321601831, 978-0321601834
Students also viewed these Physics questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



