Answered step by step
Verified Expert Solution
Question
1 Approved Answer
31 (i) Explain why the refractive index of any material with respect to air is always greater 1. (ii) In the figure below a
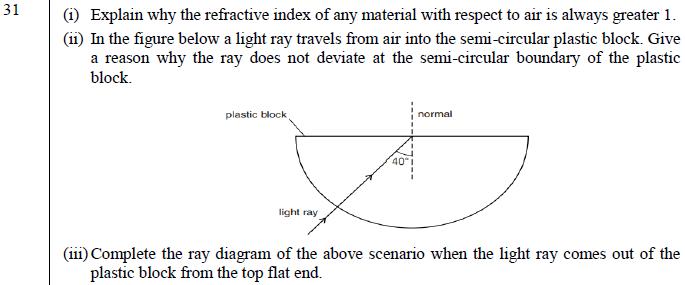




31 (i) Explain why the refractive index of any material with respect to air is always greater 1. (ii) In the figure below a light ray travels from air into the semi-circular plastic block. Give a reason why the ray does not deviate at the semi-circular boundary of the plastic block. plastic block 401 normal D light ray (iii) Complete the ray diagram of the above scenario when the light ray comes out of the plastic block from the top flat end. 32 (i) State the law that explains the heating effect of current with respect to the measurable properties in an electrical circuit. (ii) List the factors on which the resistance of a conductor depends. 33 Anannya responded to the question: Why do electrical appliances with metallic bodies are connected to the mains through a three pin plug, whereas an electric bulb can be connected with a two pin plug? She wrote: Three pin connections reduce heating of connecting wires. (i) Is her answer correct or incorrect? Justify. What is the function of a fuse in a domestic circuit? 34 a) Rehmat classified the reaction between Methane and Chlorine in presence of sunlight as a substitution reaction. Support Rehmat's view with suitable justification and illustrate the reaction with the help of a balanced chemical equation. b) Chlorine gas was prepared using electrolysis of brine solution. Write the chemical equation to represent the change. Identify the other products formed in the process and give one application of each. OR Raina while doing certain reactions observed that heating of substance 'X' with vinegar like smell with a substance 'Y' (which is used as an industrial solvent) in presence of conc. Sulphuric acid on a water bath gives a sweet-smelling liquid 'Z' having molecular formula C4H8O2. When heated with caustic soda (NaOH), 'Z' gives back the sodium salt of and the compound 'Y'. Identify 'X', 'Y', and 'Z'. Illustrate the changes with the help of suitable chemical equations. 35 Given below are certain situations. Analyze and describe its possible impact on a person: a) Testes of a male boy are not able to descend into scrotum during his embryonic development. b) Vas deferens of a man is plugged. c) Prostate and seminal vesicles are not functional. d) Egg is not fertilised in a human female. e) Placenta does not attach to the uterus optimally. OR a) A doctor has advised Sameer to reduce sugar intake in his diet and do regular exercise after checking his blood test reports. Which disease do you think Sameer is suffering from? Name the hormone responsible for this disease and the organ producing the hormone. b) Which hormone is present in the areas of rapid cell division in a plant and which hormone inhibits the growth?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


